A new study from researchers highlights an unexpected player in Alzheimer's disease: aging astrocytes.
Senescent astrocytes have been identified as a major contributor to Alzheimer's progression. The cells lose protective functions and fuel inflammation, particularly in APOE4 carriers. The findings highlight senolytic drugs as a potential new therapy.
For the first time, the research team has shown that senescent astrocytes are present in the brains of people with Alzheimer's disease. These cells remain alive but have lost their normal function, and their accumulation suggests that cellular aging plays a direct and significant role in neurodegeneration.
The findings were published in the Journal of Neuroinflammation. The study was led by Dr. Antonia Gutiérrez, Professor of Cell Biology and Principal Investigator of the NeuroAD group, alongside Dr. Juan Antonio García León, Associate Professor of Cell Biology. The research team also included Laura Cáceres, Laura Trujillo, Elba López, Elisabeth Sánchez, and Inés Moreno.
Astrocytes are the most common type of glial cell in the brain. They are essential for maintaining healthy neurons and supporting proper brain function. However, the researchers found that in individuals with the strongest genetic risk factor for Alzheimer's disease (APOE4), astrocytes show signs of abnormal early aging.
APOE4 Genotype Drives Premature Astrocyte Aging
"We have confirmed that these damaged astrocytes not only lose their ability to protect neurons but also adopt a pro-inflammatory profile that severely compromises neuronal survival," the authors explain. Upon entering a state of senescence, the cells accumulate DNA damage, exhibit mitochondrial alterations—the cellular 'powerhouses'—and 'release toxic molecules that amplify inflammation and tissue damage in the brain.
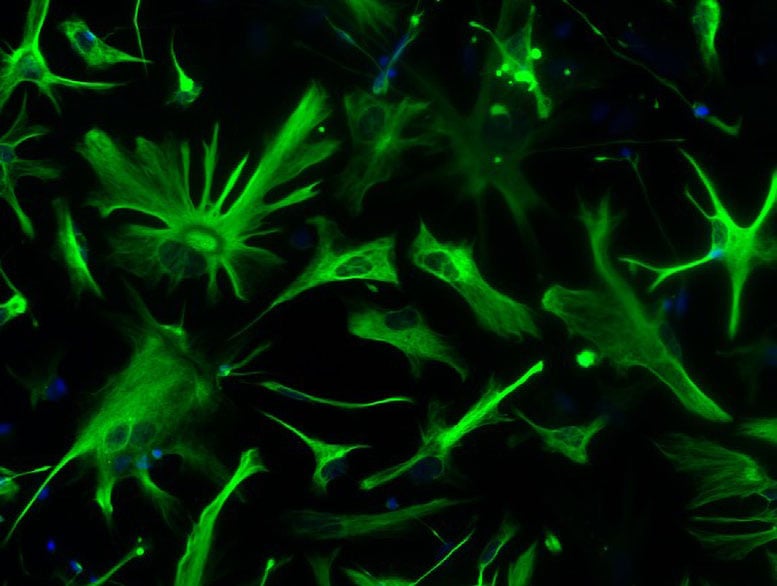
To investigate these processes, the scientists used induced pluripotent stem cells (iPSCs). By taking small skin samples from patients, they were able to reprogram the cells in the laboratory and generate functional human astrocytes for detailed study.
This approach allowed the team to examine disease mechanisms directly in human cells, avoiding some of the limitations of animal models, which do not always capture the full complexity of the human brain.
Human Validation Confirms Central Role in Neurodegeneration
The laboratory findings were reinforced by examining brain tissue obtained after death from individuals with Alzheimer's disease. The analysis strongly suggested that nearly 80 percent of the cells in the cerebral cortex that displayed signs of premature aging were astrocytes. This proportion was much higher than what is seen in healthy people of the same age.

Together, these results support the idea that astrocyte senescence is not simply a byproduct of the disease but a driving force behind its progression.
The discovery is especially important given that there is currently no effective treatment capable of curing Alzheimer's disease or stopping its advance. In Spain alone, more than 1.2 million people are affected.
The findings suggest a new therapeutic direction focused on developing innovative strategies such as senolytic drugs. These treatments would aim to eliminate or "reprogram" aged astrocytes, with the goal of preserving neurons and slowing cognitive decline.
Reference: "Human iPSC-derived APOE4/4 Alzheimer´s disease astrocytes exhibit a senescent and pro-inflammatory state that compromises neuronal support" by Laura Caceres-Palomo, Elisabeth Sanchez-Mejias, Laura Trujillo-Estrada, Juan Jose Perez-Moreno, Elba Lopez-Oliva, Tau En Lim, Leah DeFlitch, Serena H. Chang, Lucas Kampman, M. Ryan Corces, Mathew Blurton-Jones, Ines Moreno-Gonzalez, Alberto Pascual, Javier Vitorica, Juan Antonio Garcia-Leon and Antonia Gutierrez, 12 December 2025, Journal of Neuroinflammation.
DOI: 10.1186/s12974-025-03607-z
News
Scientists Rewire Natural Killer Cells To Attack Cancer Faster and Harder
Researchers tested new CAR designs in NK-92 cells and found the modified cells killed tumor cells more effectively, showing stronger anti-cancer activity. Researchers at the Ribeirão Preto Blood Center and the Center for Cell-Based [...]
New “Cellular” Target Could Transform How We Treat Alzheimer’s Disease
A new study from researchers highlights an unexpected player in Alzheimer’s disease: aging astrocytes. Senescent astrocytes have been identified as a major contributor to Alzheimer’s progression. The cells lose protective functions and fuel inflammation, particularly in [...]
Treating a Common Dental Infection… Effects That Extend Far Beyond the Mouth
Successful root canal treatment may help lower inflammation associated with heart disease and improve blood sugar and cholesterol levels. Treating an infected tooth with a successful root canal procedure may do more than relieve [...]
Microplastics found in prostate tumors in small study
In a new study, researchers found microplastics deep inside prostate cancer tumors, raising more questions about the role the ubiquitous pollutants play in public health. The findings — which come from a small study of 10 [...]
All blue-eyed people have this one thing in common
All Blue-Eyed People Have This One Thing In Common Blue Eyes Aren’t Random—Research Traces Them Back to One Prehistoric Human It sounds like a myth at first — something you’d hear in a folklore [...]
Scientists reveal how exercise protects the brain from Alzheimer’s
Researchers at UC San Francisco have identified a biological process that may explain why exercise sharpens thinking and memory. Their findings suggest that physical activity strengthens the brain's built in defense system, helping protect [...]
NanoMedical Brain/Cloud Interface – Explorations and Implications. A new book from Frank Boehm
New book from Frank Boehm, NanoappsMedical Inc Founder: This book explores the future hypothetical possibility that the cerebral cortex of the human brain might be seamlessly, safely, and securely connected with the Cloud via [...]
Deadly Pancreatic Cancer Found To “Wire Itself” Into the Body’s Nerves
A newly discovered link between pancreatic cancer and neural signaling reveals a promising drug target that slows tumor growth by blocking glutamate uptake. Pancreatic cancer is among the most deadly cancers, and scientists are [...]
This Simple Brain Exercise May Protect Against Dementia for 20 Years
A long-running study following thousands of older adults suggests that a relatively brief period of targeted brain training may have effects that last decades. Starting in the late 1990s, close to 3,000 older adults [...]
Scientists Crack a 50-Year Tissue Mystery With Major Cancer Implications
Researchers have resolved a 50-year-old scientific mystery by identifying the molecular mechanism that allows tissues to regenerate after severe damage. The discovery could help guide future treatments aimed at reducing the risk of cancer [...]
This New Blood Test Can Detect Cancer Before Tumors Appear
A new CRISPR-powered light sensor can detect the faintest whispers of cancer in a single drop of blood. Scientists have created an advanced light-based sensor capable of identifying extremely small amounts of cancer biomarkers [...]
Blindness Breakthrough? This Snail Regrows Eyes in 30 Days
A snail that regrows its eyes may hold the genetic clues to restoring human sight. Human eyes are intricate organs that cannot regrow once damaged. Surprisingly, they share key structural features with the eyes [...]
This Is Why the Same Virus Hits People So Differently
Scientists have mapped how genetics and life experiences leave lasting epigenetic marks on immune cells. The discovery helps explain why people respond so differently to the same infections and could lead to more personalized [...]
Rejuvenating neurons restores learning and memory in mice
EPFL scientists report that briefly switching on three “reprogramming” genes in a small set of memory-trace neurons restored memory in aged mice and in mouse models of Alzheimer’s disease to level of healthy young [...]
New book from Nanoappsmedical Inc. – Global Health Care Equivalency
A new book by Frank Boehm, NanoappsMedical Inc. Founder. This groundbreaking volume explores the vision of a Global Health Care Equivalency (GHCE) system powered by artificial intelligence and quantum computing technologies, operating on secure [...]
New Molecule Blocks Deadliest Brain Cancer at Its Genetic Root
Researchers have identified a molecule that disrupts a critical gene in glioblastoma. Scientists at the UVA Comprehensive Cancer Center say they have found a small molecule that can shut down a gene tied to glioblastoma, a [...]