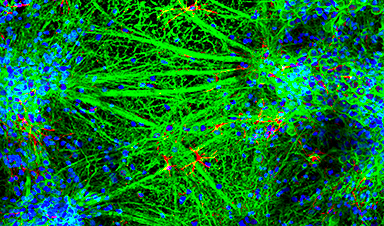
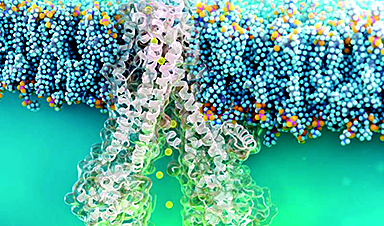
You can 3D print nearly anything: rockets, mouse ovaries, and for some reason, lamps made of orange peels. Now, scientists at Monash University in Melbourne, Australia, have printed living neural networks composed of rat brain cells that seem to mature and communicate like real brains do.
Researchers want to create mini-brains partly because they could someday offer a viable alternative to animal testing in drug trials and studies of basic brain function. At the start of 2023, the US Congress passed an annual spending bill pushing scientists to reduce their use of animals in federally funded research, following the signing of the US Food and Drug Administration's Modernization Act 2.0, which allowed high-tech alternatives in drug safety trials. Rather than testing new drugs on thousands of animals, pharmaceutical companies could apply them to 3D-printed mini-brains—in theory. There are still complexities to iron out before this moves from proof of concept to standard lab practice.
3D-printing is just one entry in the race to build a better mini-brain. One existing option is culturing a single layer of neurons in a petri dish, guiding cells to grow over recording electrodes. Growing the tissue around electrodes is convenient for running experiments, but it comes at the cost of biological realism. (Brains aren't flat.) To get closer to the brain's true structure, researchers can instead coax a bunch of stem cells to organize themselves into 3D tissues called organoids—but can't fully control how they grow.
The Monash team tried to split the difference. With 3D-printing, researchers can culture cells in specific patterns on top of recording electrodes, granting them a degree of experimental control normally reserved for flat cell cultures. But because the structure is soft enough to allow cells to migrate and reorganize themselves in 3D space, it gains some of the advantages of the organoid approach, more closely mimicking the structure of normal tissue. "You kind of have the best of both worlds," says Michael Moore, professor of biomedical engineering at Tulane University in New Orleans, Louisiana, who was not involved in this study.
Led by materials science and engineering professor John Forsythe, the Monash team described their experiment in June in Advanced Healthcare Materials. Just like how inkjet printers funnel ink from cartridges to a piece of paper, Forsythe's team printed neural structures by squeezing "bioink"—rat brain cells suspended in a gel—out of a nozzle and into a scaffold. They built their neural networks by crosshatching layer by layer, stacking eight vertical layers alternating between bioinks with and without cells. (These bioinks were extruded from different cartridges, like switching between black and color.) This structure gave cells ready access to the gel's nutrients while mimicking alternating between gray and white matter in the cortex, where gray matter contains neuron cell bodies and white matter contains the long axons connecting them.
A tiny array of microelectrodes underneath the cells recorded electrical activity in the gel surrounding the cells, while other electrodes directly stimulated the neurons and recorded their responses. Using a fluorescent dye to visualize the movement of calcium ions under a microscope, the team was able to watch the cells chemically communicate. "They behaved as we would expect," Forsythe says. "There were no surprises."
That starts by making sure you don't kill the cells when you print them. When standard 3D-printers work with plastic filaments, they melt the plastic to make it moldable, heating it up to temperatures far beyond those found in the human body. This is a nonstarter for neurons, extremely finicky cells that can survive only in carefully calibrated gels that closely replicate properties of squishy, body-temperature brains. "Making a gel that is as soft as the brain, but that you can still print through a 3D-printer, is really hard," says Moore.
"It's important not to kill the cells. But with neurons, it's really important not to kill your electrical activity," adds Stephanie Willerth, a professor of biomedical engineering at the University of Victoria in Canada, who was not involved in this study. Earlier versions of 3D-printed neural tissue often excluded glial cells, which help maintain a welcoming environment for their sensitive neuron neighbors. Without them, "neurons still have some electrical activity, but it's not going to fully replicate what you see in the body," she says.
Willerth thinks the new experiment is promising. These neural networks were made of rat cells, but "it's a proof of concept showing that you can eventually do this with human cells," Willerth says. Still, future experiments will need to replicate this level of function in human cells before these neural network models can be used in translational research and medicine.
There is also a scaling issue. The tissues printed in the Monash experiment contained a few thousand neurons per square millimeter, amounting to a couple hundred thousand cells in each 8 x 8 x 0.4 mm structure. But the human brain has about 16 billion neurons in the cortex alone, not to mention billions more glial cells.
As Moore points out, 3D-printing such delicate tissue is relatively slow, even when the final product is tiny. More work needs to be done before this precise but sluggish technique can be scaled up from academic research labs to Big Pharma, where companies are often testing dozens of drugs at once. "It's not impossible," Moore says. "It's just going to be difficult." (AxoSim, a neuroengineering startup cofounded by Moore, has already started building 3D models of human neurons and peripheral nerves for commercial drug testing.)
While this technology has the potential to replace animals in many research settings, from basic neuroscience to commercial drug development, scientists may be slow to make the switch. Often, Moore finds, scientists like him are "stuck in our ways," reluctant to spend the time, money, and effort required to move away from tried-and-true animal models. "Convincing scientists to abandon those approaches for fancy engineered tissue is going to take time," he says, "but I'm very optimistic that we will gradually reduce the number of animal studies."
When dealing with brainlike structures, one can't help but think about … thinking. While researchers don't yet have good ways of defining or measuring consciousness in lab-grown neural networks, "there are possibilities of creating living artificial neural networks using this technique," Forsythe says. Last year, a team of scientists managed to use electrical stimulation and recording to link a petri dish filled with neurons to a computer, where they appeared to learn to play Pong in about five minutes. Some, like Thomas Hartung at Johns Hopkins University, believe that 3D neural networks will merge with AI to produce "organoid intelligence" that researchers will someday be able to harness for biological computing.
In the more immediate future, Forsythe and his team hope to see how their printed neural networks fare under stress. Understanding the extent to which these tissues can regenerate after suffering cellular damage will uncover important clues about the brain's ability to heal from trauma. Someday, Forsythe believes, people may be able to receive personalized treatments for neurodegenerative diseases and other brain injuries, informed by models of their own neural tissue. Willerth envisions hospitals hosting 3D-printing suites, where future clinicians will be able to use patient biopsies to print tissues that can be used to test whether a given drug will actually work for them. "It sets the stage for that kind of personalized medicine," she says. "Papers like this will drive it forward."
Engineering personalized brain treatments will be no small feat, but the research community is well on its way. "We're inching our way closer to being able to do experiments that don't require animals in the most complex organ that we know of," says Moore. "Perhaps the most complex structure in the entire universe."
News
Bowel and Ovarian Cancers Are Dramatically Rising in Young Adults, Scientists Aren’t Sure Why
Cancer incidence is increasing, especially among younger adults, and current risk factors don’t fully account for the trend. Scientists suggest other underlying causes may be contributing. Cancer patterns in England are shifting in a [...]
New Immune Pathway Could Supercharge mRNA Cancer Vaccines
A surprising backup system in the immune response to mRNA vaccines may hold the key to more effective cancer treatments. The arrival of mRNA vaccines against SARS-CoV-2 in 2020 marked a turning point in the COVID-19 pandemic. Today, [...]
Scientists Discover “Molecular Switch” That Fuels Alzheimer’s Brain Inflammation
A newly identified trigger of brain inflammation could offer a fresh target for slowing Alzheimer’s progression. The brain has its own built-in immune system that identifies threats and responds to them. In Alzheimer’s disease, growing evidence [...]
Molecular Manufacturing: The Future of Nanomedicine – New book from NanoappsMedical Inc.
This book explores the revolutionary potential of atomically precise manufacturing technologies to transform global healthcare, as well as practically every other sector across society. This forward-thinking volume examines how envisaged Factory@Home systems might enable the cost-effective [...]
Forgotten Medicinal Plant Shows Promise in Fighting Dangerous Superbugs
A traditional medicinal plant, tormentil, shows promise against antibiotic-resistant bacteria in laboratory tests. Its compounds work by limiting bacterial growth and boosting antibiotic performance. Before the development of modern antibiotics, plant-based remedies were commonly [...]
NanoMedical Brain/Cloud Interface – Explorations and Implications. A new book from Frank Boehm
New book from Frank Boehm, NanoappsMedical Inc Founder: This book explores the future hypothetical possibility that the cerebral cortex of the human brain might be seamlessly, safely, and securely connected with the Cloud via [...]
New Research Finds Shocking Link Between Chili Peppers and Cancer
If you love spicy food, you are not alone. But scientists are taking a closer look at whether eating a lot of chili peppers could affect your cancer risk. Could your love of spicy [...]
New book from Nanoappsmedical Inc. – Global Health Care Equivalency
A new book by Frank Boehm, NanoappsMedical Inc. Founder. This groundbreaking volume explores the vision of a Global Health Care Equivalency (GHCE) system powered by artificial intelligence and quantum computing technologies, operating on secure [...]
Scientists Create “Neurobots” – Living Machines With Their Own Nervous Systems
Neurobots—xenobots with neurons—show self-organized nervous systems and enhanced behaviors, revealing new insights into how biology builds functional structures. In 2020, researchers at Tufts University developed tiny living structures known as xenobots using frog cells. These microscopic organisms [...]
Our books now available worldwide!
Online Sellers other than Amazon, Routledge, and IOPP Indigo Global Health Care Equivalency in the Age of Nanotechnology, Nanomedicine and Artifcial Intelligence Global Health Care Equivalency In The Age Of Nanotechnology, Nanomedicine And Artificial [...]
Amazonian Chocolate Could Become the Next Superfood, Scientists Say
New research into Amazonian cocoa reveals that its value may extend beyond flavor alone. Chocolate from the Amazon is already known worldwide for its distinctive taste, but new research suggests it may offer even [...]
Nanobody repairs misfolded CFTR inside cells, boosting function in cystic fibrosis
A tiny antibody component could fundamentally transform the treatment of cystic fibrosis: For the first time, researchers have succeeded in developing a so-called nanobody that penetrates directly into human cells and can repair the [...]
20-Year Study Finds Daily Multivitamins Don’t Extend Lifespan
A large, decades-long study of over 390,000 U.S. adults challenges a widespread assumption about daily multivitamins. Multivitamins are a daily habit for millions of Americans, often taken with the expectation that they will extend [...]
Novel Investment Paradigms for Regenerative Healthcare Ecosystems
Introduction The transition toward regenerative healthcare ecosystems—anchored in wellness optimization, disease prevention, eradication strategies, and healthy longevity—necessitates a structural reconfiguration of capital architectures, governance models, and incentive design. Regenerative healthcare, by definition, transcends episodic [...]
What If Consciousness Exists Beyond Your Brain
Scientists still don’t know how consciousness emerges from the brain. New ideas suggest it may not emerge at all, but instead be a basic feature of reality. Is consciousness produced by the brain, or [...]
Scientists Discover Way To Treat Lung Cancer and Its Deadly Side Effect Together
A new approach using lipid nanoparticles to deliver genetic material is showing promise in tackling two major challenges in lung cancer at once.Researchers at Oregon State University have designed a new way to tackle two of [...]