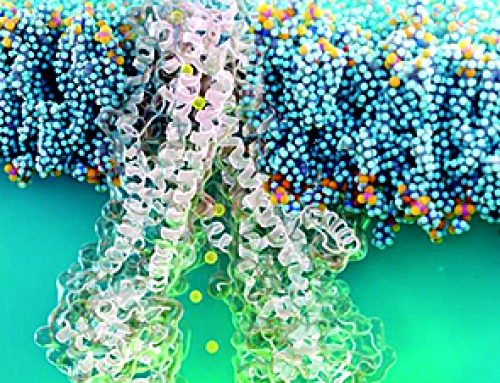
Researchers at Karolinska Institutet have in a new study used cellular reprogramming to create human three-dimensional brain models and infected these models with SARS-CoV-2. In infected models, the brain immune cells excessively eliminated synapses and acquired a gene expression pattern mimicking what has been observed in neurodegenerative disorders. The findings could help to identify new treatments against persistent cognitive symptoms after a COVID-19 infection.
In the infected models, microglia excessively engulfed synaptic structures and displayed upregulation of factors involved in phagocytosis. The developed model and the findings in the study could help to guide future efforts to target cognitive symptoms in the aftermath of COVID-19 and other neuroinvasive viral infections.
Cognitive deficits after the infection
“Interestingly, our results to a large extent mimic what has recently been observed in mouse models infected with other neuroinvasive RNA viruses such as the West Nile virus. These viruses are also linked to residual cognitive deficits after the infection, and a persisting activation of microglia leading to an excessive engulfment of synapses, which has been suggested to drive these symptoms. Multiple studies have now also reported remaining cognitive symptoms after a COVID-19 infection, as well as an increased risk of receiving a diagnosis of a disorder characterized by cognitive symptoms,” says co-first author of the study Samudyata, postdoctoral fellow in Sellgren lab at the Department of Physiology and Pharmacology at Karolinska Institutet.
Connections to Parkinson’s and Alzheimer’s disease
Microglia are the resident immune cells of the brain but also carries out important regulatory functions of the neuronal circuitries in the developing and adult brain. One of these crucial functions is to engulf unwanted synapses, a process that is believed to improve and maintain cognitive functions. However, excessive engulfment of synapses has been linked to both neurodevelopmental disorders, such as schizophrenia, as well as to neurodegenerative disorders including Alzheimer’s disease.
By sequencing genes in single cells, the authors could also study how different cell types in the model responded to the virus.
“Microglia displayed a distinct gene signature largely characterized by an upregulation of interferon-responsive genes, and included pathways previously linked to neurodegenerative disorders such as Parkinson’s and Alzheimer’s disease. This signature was also observed at a later time-point when the virus load was minimal,” says co-author of the study Susmita Malwade, doctoral student in Sellgren lab at the Department of Physiology and Pharmacology at Karolinska Institutet.
The researchers will now study how different pharmacological approaches can reverse the observed changes in the infected models.
News
Amazonian Chocolate Could Become the Next Superfood, Scientists Say
New research into Amazonian cocoa reveals that its value may extend beyond flavor alone. Chocolate from the Amazon is already known worldwide for its distinctive taste, but new research suggests it may offer even [...]
Nanobody repairs misfolded CFTR inside cells, boosting function in cystic fibrosis
A tiny antibody component could fundamentally transform the treatment of cystic fibrosis: For the first time, researchers have succeeded in developing a so-called nanobody that penetrates directly into human cells and can repair the [...]
20-Year Study Finds Daily Multivitamins Don’t Extend Lifespan
A large, decades-long study of over 390,000 U.S. adults challenges a widespread assumption about daily multivitamins. Multivitamins are a daily habit for millions of Americans, often taken with the expectation that they will extend [...]
Novel Investment Paradigms for Regenerative Healthcare Ecosystems
Introduction The transition toward regenerative healthcare ecosystems—anchored in wellness optimization, disease prevention, eradication strategies, and healthy longevity—necessitates a structural reconfiguration of capital architectures, governance models, and incentive design. Regenerative healthcare, by definition, transcends episodic [...]
What If Consciousness Exists Beyond Your Brain
Scientists still don’t know how consciousness emerges from the brain. New ideas suggest it may not emerge at all, but instead be a basic feature of reality. Is consciousness produced by the brain, or [...]
Scientists Discover Way To Treat Lung Cancer and Its Deadly Side Effect Together
A new approach using lipid nanoparticles to deliver genetic material is showing promise in tackling two major challenges in lung cancer at once.Researchers at Oregon State University have designed a new way to tackle two of [...]
Saunas Activate Your Immune System
A brief sauna session may quietly mobilize the immune system. A sauna session may do more than raise your heart rate and body temperature. A new study from Finland found that it also briefly [...]
Why music from your youth still has such an intense effect years later: A psychological perspective
You're driving, and suddenly a familiar song fills the air. Before you even know it, a wave of emotions comes over you – not just memories, but a deep, almost physical feeling. This powerful [...]
AI to antibody in days: breaking the wet lab bottleneck via high-throughput integration
The role of artificial intelligence (AI) in drug design has fundamentally shifted from a speculative tool to a central pillar of pharmaceutical research and development (R&D). Sino Biological plays a critical role in this [...]
Regenerative Healthcare by Design: Engineering Health-Centric Buildings and Urban Ecosystems
Introduction The next evolution of healthcare will not be confined to hospitals, clinics, or episodic interventions—it will be embedded into the infrastructure of everyday life. Regenerative health ecosystems require a systemic re-architecture of how [...]
Scientists Warn: Humanity Has Pushed the Planet Past Its Limits
Human population and consumption have surpassed Earth’s limits, increasing risks to climate and global stability. The Earth is already operating beyond its capacity to sustainably support the global population, according to new research highlighting [...]
Breakthrough Study Reveals Why Damaged Nerves Struggle To Heal
A newly identified molecular mechanism reveals how neurons weigh survival against repair after injury. Scientists at the Icahn School of Medicine at Mount Sinai have identified a molecular switch in neurons that limits the regrowth of [...]
Popular Vitamin B3 Supplements May Help Cancer Cells Survive, Scientists Warn
A new study raises important questions about widely used NAD+ supplements, suggesting that compounds often taken to boost energy and support healthy aging may have unintended consequences in cancer treatment. Millions of Americans take [...]
Scientists Discover Cancer Tumors Are “Addicted” to This Common Antioxidant
Cancer cells may be exploiting a common antioxidant as fuel, revealing a potential weakness that future therapies could target. Cancer cells may be tapping into an unexpected energy source: an antioxidant long associated with [...]
Nanotube injector transfers cytoplasmic contents and organelles between living cells safely
Cells are not isolated units; they continuously exchange proteins, genetic material, and even entire organelles with their neighbors. Intercellular transfer influences how tissues develop, respond to stress, and repair damage. In certain cancers, for [...]
CEO of America’s largest public hospital system is ready to replace radiologists with AI
The chief executive of America’s largest public hospital system says he is prepared to start replacing radiologists with artificial intelligence in some circumstances, once the regulatory landscape catches up. Mitchell H. Katz, MD, president [...]