The machine-learning algorithm identified a compound that kills Acinetobacter baumannii, a bacterium that lurks in many hospital settings.
Using an artificial intelligence algorithm, researchers at MIT and McMaster University have identified a new antibiotic that can kill a type of bacteria that is responsible for many drug-resistant infections.
If developed for use in patients, the drug could help to combat Acinetobacter baumannii, a species of bacteria that is often found in hospitals and can lead to pneumonia, meningitis, and other serious infections. The microbe is also a leading cause of infections in wounded soldiers in Iraq and Afghanistan.
Acinetobacter baumannii is a species of bacteria commonly found in the environment, such as soil and water, but it can also inhabit human skin and healthcare environments. It’s known for its ability to survive on artificial surfaces for extended periods, making hospitals a common site for infection. It primarily affects people with compromised immune systems, causing a range of illnesses including pneumonia, blood infections, and meningitis. A. baumannii is particularly concerning because of its resistance to many antibiotics, leading to its classification as a “superbug.” As a result, infections can be challenging to treat, posing a significant threat to healthcare settings.
The researchers identified the new drug from a library of nearly 7,000 potential drug compounds using a machine-learning model that they trained to evaluate whether a chemical compound will inhibit the growth of A. baumannii.
“This finding further supports the premise that AI can significantly accelerate and expand our search for novel antibiotics,” says James Collins, the Termeer Professor of Medical Engineering and Science in MIT’s Institute for Medical Engineering and Science (IMES) and Department of Biological Engineering. “I’m excited that this work shows that we can use AI to help combat problematic pathogens such as A. baumannii.”
Collins and Stokes are the senior authors of the new study, which was published on May 25 in the journal Nature Chemical Biology. The paper’s lead authors are McMaster University graduate students Gary Liu and Denise Catacutan and recent McMaster graduate Khushi Rathod.
Drug discovery
Over the past several decades, many pathogenic bacteria have become increasingly resistant to existing antibiotics, while very few new antibiotics have been developed.
Several years ago, Collins, Stokes, and MIT Professor Regina Barzilay (who is also an author on the new study), set out to combat this growing problem by using machine learning, a type of artificial intelligence that can learn to recognize patterns in vast amounts of data. Collins and Barzilay, who co-direct MIT’s Abdul Latif Jameel Clinic for Machine Learning in Health, hoped this approach could be used to identify new antibiotics whose chemical structures are different from any existing drugs.
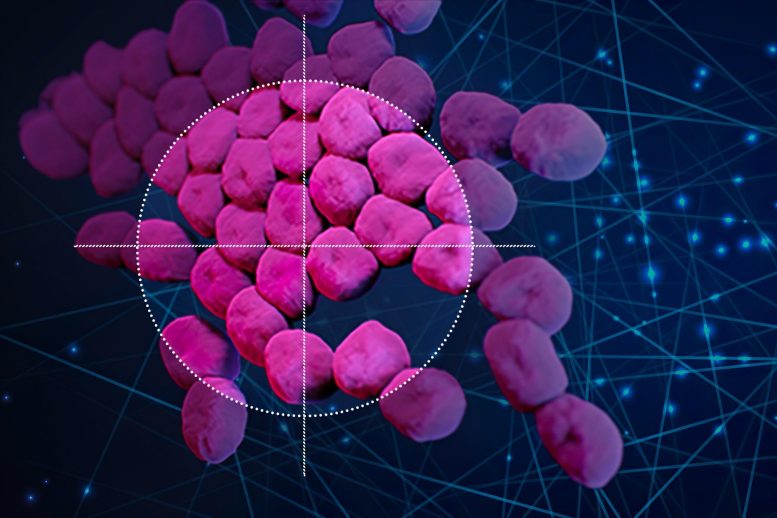
Using an artificial intelligence algorithm, researchers at MIT and McMaster University have identified a new antibiotic that can kill a type of bacteria (Acinetobacter baumannii, pink) that is responsible for many drug-resistant infections. Credit: Christine Daniloff/MIT; Acinetobacter baumannii image courtesy of CDC
In their initial demonstration, the researchers trained a machine-learning algorithm to identify chemical structures that could inhibit growth of E. coli. In a screen of more than 100 million compounds, that algorithm yielded a molecule that the researchers called halicin, after the fictional artificial intelligence system from “2001: A Space Odyssey.” This molecule, they showed, could kill not only E. coli but several other bacterial species that are resistant to treatment.
“After that paper, when we showed that these machine-learning approaches can work well for complex antibiotic discovery tasks, we turned our attention to what I perceive to be public enemy No. 1 for multidrug-resistant bacterial infections, which is Acinetobacter,” Stokes says.
To obtain training data for their computational model, the researchers first exposed A. baumannii grown in a lab dish to about 7,500 different chemical compounds to see which ones could inhibit growth of the microbe. Then they fed the structure of each molecule into the model. They also told the model whether each structure could inhibit bacterial growth or not. This allowed the algorithm to learn chemical features associated with growth inhibition.
Once the model was trained, the researchers used it to analyze a set of 6,680 compounds it had not seen before, which came from the Drug Repurposing Hub at the Broad Institute. This analysis, which took less than two hours, yielded a few hundred top hits. Of these, the researchers chose 240 to test experimentally in the lab, focusing on compounds with structures that were different from those of existing antibiotics or molecules from the training data.
Those tests yielded nine antibiotics, including one that was very potent. This compound, which was originally explored as a potential diabetes drug, turned out to be extremely effective at killing A. baumannii but had no effect on other species of bacteria including Pseudomonas aeruginosa, Staphylococcus aureus, and carbapenem-resistant Enterobacteriaceae.
This “narrow spectrum” killing ability is a desirable feature for antibiotics because it minimizes the risk of bacteria rapidly spreading resistance against the drug. Another advantage is that the drug would likely spare the beneficial bacteria that live in the human gut and help to suppress opportunistic infections such as Clostridium difficile.
“Antibiotics often have to be administered systemically, and the last thing you want to do is cause significant dysbiosis and open up these already sick patients to secondary infections,” Stokes says.
A novel mechanism
In studies in mice, the researchers showed that the drug, which they named abaucin, could treat wound infections caused by A. baumannii. They also showed, in lab tests, that it works against a variety of drug-resistant A. baumannii strains isolated from human patients.
Further experiments revealed that the drug kills cells by interfering with a process known as lipoprotein trafficking, which cells use to transport proteins from the interior of the cell to the cell envelope. Specifically, the drug appears to inhibit LolE, a protein involved in this process.
All Gram-negative bacteria express this enzyme, so the researchers were surprised to find that abaucin is so selective in targeting A. baumannii. They hypothesize that slight differences in how A. baumannii performs this task might account for the drug’s selectivity.
“We haven’t finalized the experimental data acquisition yet, but we think it’s because A. baumannii does lipoprotein trafficking a little bit differently than other Gram-negative species. We believe that’s why we’re getting this narrow spectrum activity,” Stokes says.
Stokes’ lab is now working with other researchers at McMaster to optimize the medicinal properties of the compound, in hopes of developing it for eventual use in patients.
The researchers also plan to use their modeling approach to identify potential antibiotics for other types of drug-resistant infections, including those caused by Staphylococcus aureus and Pseudomonas aeruginosa.
News
New Once-a-Week Shot Promises Life-Changing Relief for Parkinson’s Patients
A once-a-week shot from Australian scientists could spare people with Parkinson’s the grind of taking pills several times a day. The tiny, biodegradable gel sits under the skin and releases steady doses of two [...]
Weekly injectable drug offers hope for Parkinson’s patients
A new weekly injectable drug could transform the lives of more than eight million people living with Parkinson's disease, potentially replacing the need for multiple daily tablets. Scientists from the University of South Australia [...]
Most Plastic in the Ocean Is Invisible—And Deadly
Nanoplastics—particles smaller than a human hair—can pass through cell walls and enter the food web. New research suggest 27 million metric tons of nanoplastics are spread across just the top layer of the North [...]
Repurposed drugs could calm the immune system’s response to nanomedicine
An international study led by researchers at the University of Colorado Anschutz Medical Campus has identified a promising strategy to enhance the safety of nanomedicines, advanced therapies often used in cancer and vaccine treatments, [...]
Nano-Enhanced Hydrogel Strategies for Cartilage Repair
A recent article in Engineering describes the development of a protein-based nanocomposite hydrogel designed to deliver two therapeutic agents—dexamethasone (Dex) and kartogenin (KGN)—to support cartilage repair. The hydrogel is engineered to modulate immune responses and promote [...]
New Cancer Drug Blocks Tumors Without Debilitating Side Effects
A new drug targets RAS-PI3Kα pathways without harmful side effects. It was developed using high-performance computing and AI. A new cancer drug candidate, developed through a collaboration between Lawrence Livermore National Laboratory (LLNL), BridgeBio Oncology [...]
Scientists Are Pretty Close to Replicating the First Thing That Ever Lived
For 400 million years, a leading hypothesis claims, Earth was an “RNA World,” meaning that life must’ve first replicated from RNA before the arrival of proteins and DNA. Unfortunately, scientists have failed to find [...]
Why ‘Peniaphobia’ Is Exploding Among Young People (And Why We Should Be Concerned)
An insidious illness is taking hold among a growing proportion of young people. Little known to the general public, peniaphobia—the fear of becoming poor—is gaining ground among teens and young adults. Discover the causes [...]
Team finds flawed data in recent study relevant to coronavirus antiviral development
The COVID pandemic illustrated how urgently we need antiviral medications capable of treating coronavirus infections. To aid this effort, researchers quickly homed in on part of SARS-CoV-2's molecular structure known as the NiRAN domain—an [...]
Drug-Coated Neural Implants Reduce Immune Rejection
Summary: A new study shows that coating neural prosthetic implants with the anti-inflammatory drug dexamethasone helps reduce the body’s immune response and scar tissue formation. This strategy enhances the long-term performance and stability of electrodes [...]
Scientists discover cancer-fighting bacteria that ‘soak up’ forever chemicals in the body
A family of healthy bacteria may help 'soak up' toxic forever chemicals in the body, warding off their cancerous effects. Forever chemicals, also known as PFAS (per- and polyfluoroalkyl substances), are toxic chemicals that [...]
Johns Hopkins Researchers Uncover a New Way To Kill Cancer Cells
A new study reveals that blocking ribosomal RNA production rewires cancer cell behavior and could help treat genetically unstable tumors. Researchers at the Johns Hopkins Kimmel Cancer Center and the Department of Radiation Oncology and Molecular [...]
AI matches doctors in mapping lung tumors for radiation therapy
In radiation therapy, precision can save lives. Oncologists must carefully map the size and location of a tumor before delivering high-dose radiation to destroy cancer cells while sparing healthy tissue. But this process, called [...]
Scientists Finally “See” Key Protein That Controls Inflammation
Researchers used advanced microscopy to uncover important protein structures. For the first time, two important protein structures in the human body are being visualized, thanks in part to cutting-edge technology at the University of [...]
AI tool detects 9 types of dementia from a single brain scan
Mayo Clinic researchers have developed a new artificial intelligence (AI) tool that helps clinicians identify brain activity patterns linked to nine types of dementia, including Alzheimer's disease, using a single, widely available scan—a transformative [...]
Is plastic packaging putting more than just food on your plate?
New research reveals that common food packaging and utensils can shed microscopic plastics into our food, prompting urgent calls for stricter testing and updated regulations to protect public health. Beyond microplastics: The analysis intentionally [...]