In efforts to tackle the leading cause of blindness in developed countries, researchers have recruited nanotechnology to help regrow retinal cells.
Macular degeneration is a form of central vision loss, which has massive social, mobility, and mental consequences. It impacts hundreds of millions of people globally and is increasing in prevalence.
The degeneration is the consequence of damaged retinal pigment cells. Our bodies are unable to grow and replace these cells once they start dying, so scientists have been exploring alternative methods to replace them and the membrane within which they sit.
"In the past, scientists would grow cells on a flat surface, which is not biologically relevant," explains Anglia Ruskin University biochemist Barbara Pierscionek.
"Using these new techniques the cell line has been shown to thrive in the 3D environment provided by the scaffolds."
Nottingham Trent University biomedical scientist Biola Egbowon and colleagues fabricated these 3D scaffolds with polymer nanofibers and coated them with a steroid to reduce inflammation.
Using a technique called electrospinning, which produces nanometer-wide fibers by squirting a molten polymer through a high-voltage field, the team was able to keep the scaffold sufficiently thin.
The polyacrylonitrile polymer they used provided mechanical strength, and Jeffamine polymer attracts water, essentially allowing the synthetic scaffold to act as a membrane.
The water-attracting ability of the material is what helps the cells bind to the scaffold and also encourages their growth, but when the effect is too strong, it's also been associated with cell death in previous research.
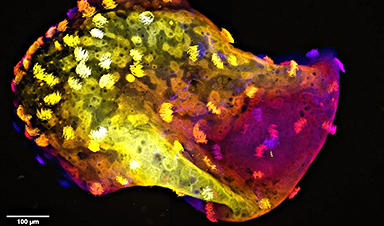
The team's new formulation seems to be just right, as the system increased the growth and longevity of the retinal lab cells and kept them viable for at least 150 days.
"This research has demonstrated, for the first time, that nanofiber scaffolds treated with the anti-inflammatory substance such as fluocinolone acetonide can enhance the growth, differentiation, and functionality of retinal pigment epithelial cells," says Pierscionek.
Previous attempts have used collagen and cellulose to create a similar scaffold, but Egbowon and team believes their synthetic option will be easier to make compatible with our immune systems and simpler to modify.
The new study has demonstrated this method can keep the required single layer of retinal cells healthy, producing biomarkers that indicate they are functioning more naturally than what has been found when they grow on other mediums.
However, there's still a lot we don't know about how viable this approach will be for treating human patients with macular degeneration.
"While this may indicate the potential of such cellularized scaffolds in regenerative medicine, it does not address the question of biocompatibility with human tissue," Egbowon and colleagues caution in their paper, as there is a massive difference between growing cells in a petri dish and having a functioning tissue substitute within a body.
Other research in this area is already investigating whether lab grown cells can be plugged back into other retinal cell types to form functioning units of tissue. Another tactic involves activating cells already in human eye tissues that regenerate retinal cells in other animals.
The team's next steps will be to investigate the orientation of the cells, which is important for ensuring they can maintain a good blood supply, before they can be considered for testing inside a living system.
This research was published in Materials & Design.
News
New Immune Pathway Could Supercharge mRNA Cancer Vaccines
A surprising backup system in the immune response to mRNA vaccines may hold the key to more effective cancer treatments. The arrival of mRNA vaccines against SARS-CoV-2 in 2020 marked a turning point in the COVID-19 pandemic. Today, [...]
Scientists Discover “Molecular Switch” That Fuels Alzheimer’s Brain Inflammation
A newly identified trigger of brain inflammation could offer a fresh target for slowing Alzheimer’s progression. The brain has its own built-in immune system that identifies threats and responds to them. In Alzheimer’s disease, growing evidence [...]
Molecular Manufacturing: The Future of Nanomedicine – New book from NanoappsMedical Inc.
This book explores the revolutionary potential of atomically precise manufacturing technologies to transform global healthcare, as well as practically every other sector across society. This forward-thinking volume examines how envisaged Factory@Home systems might enable the cost-effective [...]
Forgotten Medicinal Plant Shows Promise in Fighting Dangerous Superbugs
A traditional medicinal plant, tormentil, shows promise against antibiotic-resistant bacteria in laboratory tests. Its compounds work by limiting bacterial growth and boosting antibiotic performance. Before the development of modern antibiotics, plant-based remedies were commonly [...]
NanoMedical Brain/Cloud Interface – Explorations and Implications. A new book from Frank Boehm
New book from Frank Boehm, NanoappsMedical Inc Founder: This book explores the future hypothetical possibility that the cerebral cortex of the human brain might be seamlessly, safely, and securely connected with the Cloud via [...]
New Research Finds Shocking Link Between Chili Peppers and Cancer
If you love spicy food, you are not alone. But scientists are taking a closer look at whether eating a lot of chili peppers could affect your cancer risk. Could your love of spicy [...]
New book from Nanoappsmedical Inc. – Global Health Care Equivalency
A new book by Frank Boehm, NanoappsMedical Inc. Founder. This groundbreaking volume explores the vision of a Global Health Care Equivalency (GHCE) system powered by artificial intelligence and quantum computing technologies, operating on secure [...]
Scientists Create “Neurobots” – Living Machines With Their Own Nervous Systems
Neurobots—xenobots with neurons—show self-organized nervous systems and enhanced behaviors, revealing new insights into how biology builds functional structures. In 2020, researchers at Tufts University developed tiny living structures known as xenobots using frog cells. These microscopic organisms [...]
Our books now available worldwide!
Online Sellers other than Amazon, Routledge, and IOPP Indigo Global Health Care Equivalency in the Age of Nanotechnology, Nanomedicine and Artifcial Intelligence Global Health Care Equivalency In The Age Of Nanotechnology, Nanomedicine And Artificial [...]
Amazonian Chocolate Could Become the Next Superfood, Scientists Say
New research into Amazonian cocoa reveals that its value may extend beyond flavor alone. Chocolate from the Amazon is already known worldwide for its distinctive taste, but new research suggests it may offer even [...]
Nanobody repairs misfolded CFTR inside cells, boosting function in cystic fibrosis
A tiny antibody component could fundamentally transform the treatment of cystic fibrosis: For the first time, researchers have succeeded in developing a so-called nanobody that penetrates directly into human cells and can repair the [...]
20-Year Study Finds Daily Multivitamins Don’t Extend Lifespan
A large, decades-long study of over 390,000 U.S. adults challenges a widespread assumption about daily multivitamins. Multivitamins are a daily habit for millions of Americans, often taken with the expectation that they will extend [...]
Novel Investment Paradigms for Regenerative Healthcare Ecosystems
Introduction The transition toward regenerative healthcare ecosystems—anchored in wellness optimization, disease prevention, eradication strategies, and healthy longevity—necessitates a structural reconfiguration of capital architectures, governance models, and incentive design. Regenerative healthcare, by definition, transcends episodic [...]
What If Consciousness Exists Beyond Your Brain
Scientists still don’t know how consciousness emerges from the brain. New ideas suggest it may not emerge at all, but instead be a basic feature of reality. Is consciousness produced by the brain, or [...]
Scientists Discover Way To Treat Lung Cancer and Its Deadly Side Effect Together
A new approach using lipid nanoparticles to deliver genetic material is showing promise in tackling two major challenges in lung cancer at once.Researchers at Oregon State University have designed a new way to tackle two of [...]
Saunas Activate Your Immune System
A brief sauna session may quietly mobilize the immune system. A sauna session may do more than raise your heart rate and body temperature. A new study from Finland found that it also briefly [...]