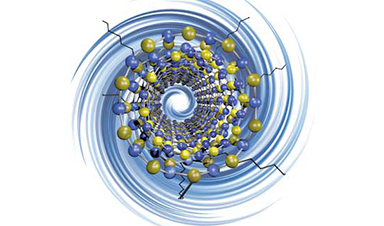
Boron nitride nanotubes are primed to become effective building blocks for next-generation composite and polymer materials based on a new discovery at Rice University – and a previous one.
Scientists at known-for-nano Rice have found a way to enhance a unique class of nanotubes using a chemical process pioneered at the university. The Rice lab of chemist Angel Martí took advantage of the Billups-Birch reaction process to enhance boron nitride nanotubes.
The work is described in the American Chemical Society journal ACS Applied Nano Materials (“Chemical Decoration of Boron Nitride Nanotubes Using the Billups-Birch Reaction: Toward Enhanced Thermostable Reinforced Polymer and Ceramic Nanocomposites”).
Boron nitride nanotubes, like their carbon cousins, are rolled sheets of hexagonal arrays. Unlike carbon nanotubes, they’re electrically insulating hybrids made of alternating boron and nitrogen atoms.
Insulating nanotubes that can be functionalized will be a valuable building block for nanoengineering projects, Martí said. “Carbon nanotubes have outstanding properties, but you can only get them in semiconducting or metallic conducting types,” he said. “Boron nitride nanotubes are complementary materials that can fill that gap.”
Until now, these nanotubes have steadfastly resisted functionalization, the “decorating” of structures with chemical additives that allows them to be customized for applications. The very properties that give boron nitride nanotubes strength and stability, especially at high temperatures, also make them hard to modify for their use in the production of advanced materials.
But the Billups-Birch reaction developed by Rice Professor Emeritus of Chemistry Edward Billups, which frees electrons to bind with other atoms, allowed Martí and lead author Carlos de los Reyes to give the electrically inert boron nitride nanotubes a negative charge.
That, in turn, opened them up to functionalization with other small molecules, including aliphatic carbon chains.
“Functionalizing the nanotubes modifies or tunes their properties,” Martí said. “When they’re pristine they are dispersible in water, but once we attach these alkyl chains, they are extremely hydrophobic (water-avoiding). Then, if you put them in very hydrophobic solvents like those with long-chain hydrocarbons, they are more dispersible than their pristine form.
Image Credit: Martí Research Group
News This Week
Why More People in Their 30s Are Suddenly Getting Colon Cancer
A major Swiss study found that colorectal cancer is becoming increasingly common in adults under 50, even as rates decline in older age groups. Researchers in Switzerland have identified a concerning trend: while colorectal [...]
Researchers Compare MS Models to Human Tissue in Search for Better Therapies
Researchers identified key differences between two widely used multiple sclerosis models, showing how each can better study myelin damage, immune responses, and repair. The findings may improve efforts to develop treatments that restore lost [...]
Scientists Discover Genetic “Off Switch” That Supercharges CAR T Cells Against Cancer
A new study reveals a possible way to make CAR T-cell therapy more durable and effective by targeting a single gene-regulating protein. CAR T-cell therapy is widely seen as a breakthrough in personalized cancer [...]
New Vitamin B12-Based Therapy Could Change How Brain Cancer Is Treated
Researchers have identified a vitamin B12–based compound that appears capable of crossing the blood–brain barrier and selectively accumulating in glioblastoma tissue. For decades, one of the biggest problems in brain cancer treatment has had [...]
Simple Fiber Supplement Cuts Knee Arthritis Pain in Just 6 Weeks, Study Finds
A daily inulin supplement may help reduce knee osteoarthritis pain while revealing a possible link between gut health, muscle function, and pain sensitivity. For millions of people living with knee osteoarthritis, managing chronic pain [...]
This Common Vitamin May Help Stop Prediabetes From Turning Into Diabetes
Vitamin D may help prevent type 2 diabetes in people with specific genetic variations, offering a possible path toward personalized diabetes prevention. More than 40% of U.S. adults have prediabetes, a condition in which [...]
Ebola, hantavirus: Is the world prepared for the next pandemic?
Funding cuts to health research and a growing antivaccine movement are making it harder than ever to respond to viruses. The World Health Organization (WHO) has declared that an Ebola outbreak in Uganda and [...]
May 2026 Healthcare News and Trends: Market Signals That Matter
Artificial intelligence is dominating headlines, telehealth has settled into a new normal, and digital health continues to promise transformation. However, much of what is being discussed in healthcare today reflects potential rather than reality. [...]



Leave A Comment