Fragile mRNA molecules used in COVID-19 vaccines can’t get into cells on their own. They owe their success to lipid nanoparticles that took decades to refine.
Messenger RNA (mRNA) is having a moment. This year, hundreds of millions of people will receive shots of the Pfizer-BioNTech or Moderna vaccines for COVID-19. The crucial ingredient in each injection is mRNA, short-lived strands of genetic material that prompt our cells to start making SARS-CoV-2 proteins, which in turn help our immune systems develop antibodies that prevent future infections. Thanks to decades of scientific perseverance, billions of dollars of investment in the technology, and previous work on coronaviruses, the vaccine makers were able to design their vaccines and prove their safety and efficacy in under a year.
The success of these COVID-19 vaccines is remarkable and was far from guaranteed. mRNA is incredibly delicate. Enzymes in the environment and in our bodies are quick to chop mRNA into pieces, making lab experiments difficult and the delivery of mRNA to our cells daunting. On top of that, mRNA strands are large and negatively charged and can’t simply waltz across the protective lipid membranes of cells. Many scientists thought the technology would never work.
“There were many, many skeptics,” says Frank DeRosa, who began working with mRNA in 2008 and is now chief technology officer at Translate Bio, a firm developing mRNA vaccines with Sanofi. “People used to say that if you looked at it wrong it would fall apart.”
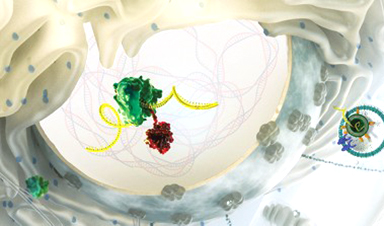
Luckily, scientists found a solution. To protect the fragile molecule as it sneaks into cells, they turned to a delivery technology with origins older than the idea of mRNA therapy itself: tiny balls of fat called lipid nanoparticles, or LNPs.
LNPs used in the COVID-19 vaccines contain just four ingredients: ionizable lipids whose positive charges bind to the negatively charged backbone of mRNA, pegylated lipids that help stabilize the particle, and phospholipids and cholesterol molecules that contribute to the particle’s structure. Thousands of these four components encapsulate mRNA, shield it from destructive enzymes, and shuttle it into cells, where the mRNA is unloaded and used to make proteins. Although the concept seems simple, perfecting it was far from straightforward.
It is a tremendous vindication for everyone working in controlled drug delivery.Robert Langer, chemical engineer, Massachusetts Institute of Technology
Over more than 3 decades, promising lipids studied in the lab often failed to live up to their potential when tested in animals or humans. Positively charged lipids are inherently toxic, and companies struggled for years before landing on formulations that were safe and effective. When injected intravenously, the particles invariably accumulated in the liver, and delivery to other organs is still an obstacle. Reliably manufacturing consistent LNPs was another challenge, and producing the raw materials needed to make the particles is a limiting factor in the production of COVID-19 vaccines today.
LNP development has been a headache, but without this packaging, mRNA vaccines would be nothing. “It is the unsung hero of the whole thing,” says Giuseppe Ciaramella, who was head of infectious diseases at Moderna from 2014 to 2018…..
News
UCLA Scientists Uncover a “Hidden Weakness” in Some of the World’s Deadliest Cancers
A new study has uncovered an unexpected vulnerability in some of the deadliest cancers. Researchers at UCLA have identified a previously hidden weakness in some of the most aggressive cancers, pointing to a possible new way [...]
AI-designed universal coronavirus vaccine clears first human trial
Key Takeaways Super-Antigen Technology: Uses AI and machine learning to analyze viral genomes, creating a single vaccine that targets essential features across entire virus families, including coronaviruses and Ebola. Human Trials & Safety: Phase [...]
Researchers Discover a Hidden Vitamin D Problem That Persists Year-Round
A new study suggests that some groups may not experience the expected seasonal boost in vitamin D levels, even during the sunniest months of the year. Many people assume that spending more time outdoors [...]
Researchers Solve the Mystery Behind a Billion-Dollar Dental Implant Disease
Researchers have uncovered why a common and costly dental implant infection often resists antibiotics. Dental implants have helped tens of millions of people regain a full set of stable, functional teeth, something traditional dentures [...]
Nanoparticles inspired by lung fluid improve therapies targeting respiratory system
The CIC biomaGUNE Center for Cooperative Research in Biomaterials has developed pulmonary surfactant nanoparticles (the blend of lipids and proteins that line the alveoli and enables breathing), which are encapsulated [...]
Scientists Finally Uncover How a “Forever Chemical” Causes Birth Defects
PFDA, a PFAS “forever chemical,” can cause craniofacial birth defects by disrupting retinoic acid regulation during fetal development, revealing the first clear molecular mechanism behind the link. Researchers have long linked perfluoroalkyl and polyfluoroalkyl substances (PFAS), [...]
Scientists Have Discovered These Deadly Parasites Are Secretly Swapping DNA
Leishmania parasites appear to evolve through widespread genetic exchange, reshaping assumptions about how they adapt and spread. A parasite long thought to spread mostly by cloning itself may be far more genetically dynamic than [...]
Stanford’s Revolutionary New Microscope Reveals Living Cells in Stunning Detail
Stanford researchers have developed a microscope that can show how nanostructures interact inside living cells at the highest resolution achieved so far. The view into living cells just got better. Stanford researchers have merged [...]
What Bundibugyo Ebola vaccines and treatments are under development
By Mariam Sunny and Jennifer Rigby May 29 (Reuters) – Global health authorities are racing to identify medical options to help contain an Ebola outbreak in eastern Democratic Republic of Congo, linked to the [...]
Why More People in Their 30s Are Suddenly Getting Colon Cancer
A major Swiss study found that colorectal cancer is becoming increasingly common in adults under 50, even as rates decline in older age groups. Researchers in Switzerland have identified a concerning trend: while colorectal [...]
Researchers Compare MS Models to Human Tissue in Search for Better Therapies
Researchers identified key differences between two widely used multiple sclerosis models, showing how each can better study myelin damage, immune responses, and repair. The findings may improve efforts to develop treatments that restore lost [...]
Scientists Discover Genetic “Off Switch” That Supercharges CAR T Cells Against Cancer
A new study reveals a possible way to make CAR T-cell therapy more durable and effective by targeting a single gene-regulating protein. CAR T-cell therapy is widely seen as a breakthrough in personalized cancer [...]
New Vitamin B12-Based Therapy Could Change How Brain Cancer Is Treated
Researchers have identified a vitamin B12–based compound that appears capable of crossing the blood–brain barrier and selectively accumulating in glioblastoma tissue. For decades, one of the biggest problems in brain cancer treatment has had [...]
Simple Fiber Supplement Cuts Knee Arthritis Pain in Just 6 Weeks, Study Finds
A daily inulin supplement may help reduce knee osteoarthritis pain while revealing a possible link between gut health, muscle function, and pain sensitivity. For millions of people living with knee osteoarthritis, managing chronic pain [...]
This Common Vitamin May Help Stop Prediabetes From Turning Into Diabetes
Vitamin D may help prevent type 2 diabetes in people with specific genetic variations, offering a possible path toward personalized diabetes prevention. More than 40% of U.S. adults have prediabetes, a condition in which [...]
Ebola, hantavirus: Is the world prepared for the next pandemic?
Funding cuts to health research and a growing antivaccine movement are making it harder than ever to respond to viruses. The World Health Organization (WHO) has declared that an Ebola outbreak in Uganda and [...]