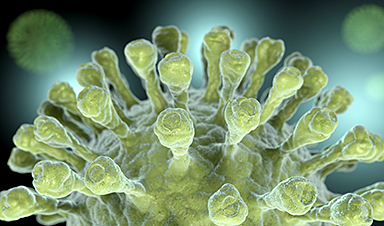
A coronavirus uses protein “spikes” to grab and infect cells. Despite their name, those spikes aren’t stiff and pointy. They’re shaped like chicken drumsticks with the meaty part facing out, and the meaty part can tilt every which way on its slender stalk. That ability to tilt, it turns out, affects how successfully the spike can infect a cell.
While the study was carried out on a much less dangerous cousin of SARS-CoV-2, the coronavirus that causes COVID-19, it has implications for COVID-19, too, since both viruses bind to the same receptor on a cell’s surface to initiate infection, said Jing Jin, a biologist at Vitalant Research Institute and adjunct assistant professor at the University of California, San Francisco who performed virology experiments for the study.
The results, she said, suggest that disabling the spike’s hinges could be a good way to prevent or treat a wide range of coronavirus infections.
The team also discovered that each coronavirus particle is unique, both in its underlying shape and its display of spikes. Some are spherical, some are not; some bristle with spikes while others are nearly bald.
“The spikes are floppy and move around, and we used a combination of tools to explore all their possible angles and orientations,” said Greg Pintilie, a Stanford scientist who developed detailed 3D models of the virus and its spikes. Seen up close, he said, each spike is different from all the rest, mainly in its direction and degree of tilting.
The research team reported its findings in Nature Communications.
“Since the pandemic started, most studies have looked at the structures of coronavirus spike proteins that were not attached to the virus itself,” said Wah Chiu, a professor at SLAC and Stanford and co-director of the Stanford-SLAC Cryo-EM facilities where the imaging was done. “These are the first images made of the spikes of this strain of coronavirus while they’re still attached to the virus particles.”
SARS-CoV-2’s more benign cousin
The study has roots in the early days of the pandemic, when research at SLAC shut down except for work aimed at understanding, preventing and treating COVID-19 infections.
Because experiments with the actual SARS-CoV-2 virus can only take place in high-level (BSL3) biosafety labs, many scientists chose to work with more benign members of the coronavirus family. Chiu and his colleagues selected human coronavirus NL63 as their subject. It causes up to 10% of human respiratory infections, mainly in children and immunocompromised people, with symptoms ranging from mild coughs and sniffles to bronchitis and croup.
In 2020, Chiu said, the team used cryogenic electron microscopy (cryo-EM) and computational analysis to image the crowns of NL63 spikes with near-atomic resolution.
But because a spike’s stalk is much thinner than its crown, they were not able to get clear, high-resolution images of both at once.
Zooming in on spikes
This study combined information gleaned from a series of experiments to get a much more complete picture.
First, Stanford graduate student David Chmielewski used cryogenic electron tomography (cryo-ET) to combine cryo-EM images of viruses that were taken from different angles into high-resolution 3D images of more than a hundred NL63 particles.
SLAC senior scientist Michael Schmid plugged those images into a 3D visualization tool and discovered that each of a particle’s spikes was bent in a unique way. Another SLAC scientist, Muyuan Chen, used advanced image reconstruction to create maps showing the average density of the spikes’ crowns and stalks.
Zooming in on one of those spikes, biological chemist Lance Wells at the University of Georgia used a technique called mass spectrometry to pinpoint the site-specific chemical compositions of the 39 sugar chains attached to each of the spike’s three identical proteins.
Finally, Abhishek Singharoy, a computational biophysicist at Arizona State University, and his student, Eric Wilson, integrated all those measurements into atomic models of the spikes’ crowns and stalks at different bending angles, and carried out further simulations to see how far and how freely a spike can bend.
“It turns out that no matter what, the spikes have a preferred bending angle of about 50 degrees,” Chiu said, “and they can tilt up to 80 degrees in any direction in the simulation, which matches well with our cryo-ET experimental observations.”
The bending occurred at a place on the stalk, just below the crown, where a particular cluster of sugar molecules clung to the protein, forming a hinge. Computer simulations suggested that changes in the structure of this hinge would affect its ability to bend, and lab experiments went one step further: They showed that mutations in the protein part of the hinge made the spike much less infectious. This suggests that targeting the hinge could provide an avenue to fight the virus.
“People working on the more dangerous coronaviruses, including MERS-CoV and SARS-CoV-2, have identified a region equivalent to this one and discovered antibodies targeting this region,” Jin said. “That tells us it’s a critical region that is highly conserved, meaning that it has stayed much the same over the course of evolution. So maybe by targeting this region in all coronaviruses, we can come up with a universal therapy or vaccine.”
More information: David Chmielewski et al, Structural insights into the modulation of coronavirus spike tilting and infectivity by hinge glycans, Nature Communications (2023). DOI: 10.1038/s41467-023-42836-9
News
20-Year Study Finds Daily Multivitamins Don’t Extend Lifespan
A large, decades-long study of over 390,000 U.S. adults challenges a widespread assumption about daily multivitamins. Multivitamins are a daily habit for millions of Americans, often taken with the expectation that they will extend [...]
Novel Investment Paradigms for Regenerative Healthcare Ecosystems
Introduction The transition toward regenerative healthcare ecosystems—anchored in wellness optimization, disease prevention, eradication strategies, and healthy longevity—necessitates a structural reconfiguration of capital architectures, governance models, and incentive design. Regenerative healthcare, by definition, transcends episodic [...]
What If Consciousness Exists Beyond Your Brain
Scientists still don’t know how consciousness emerges from the brain. New ideas suggest it may not emerge at all, but instead be a basic feature of reality. Is consciousness produced by the brain, or [...]
Scientists Discover Way To Treat Lung Cancer and Its Deadly Side Effect Together
A new approach using lipid nanoparticles to deliver genetic material is showing promise in tackling two major challenges in lung cancer at once.Researchers at Oregon State University have designed a new way to tackle two of [...]
Saunas Activate Your Immune System
A brief sauna session may quietly mobilize the immune system. A sauna session may do more than raise your heart rate and body temperature. A new study from Finland found that it also briefly [...]
Why music from your youth still has such an intense effect years later: A psychological perspective
You're driving, and suddenly a familiar song fills the air. Before you even know it, a wave of emotions comes over you – not just memories, but a deep, almost physical feeling. This powerful [...]
AI to antibody in days: breaking the wet lab bottleneck via high-throughput integration
The role of artificial intelligence (AI) in drug design has fundamentally shifted from a speculative tool to a central pillar of pharmaceutical research and development (R&D). Sino Biological plays a critical role in this [...]
Regenerative Healthcare by Design: Engineering Health-Centric Buildings and Urban Ecosystems
Introduction The next evolution of healthcare will not be confined to hospitals, clinics, or episodic interventions—it will be embedded into the infrastructure of everyday life. Regenerative health ecosystems require a systemic re-architecture of how [...]
Scientists Warn: Humanity Has Pushed the Planet Past Its Limits
Human population and consumption have surpassed Earth’s limits, increasing risks to climate and global stability. The Earth is already operating beyond its capacity to sustainably support the global population, according to new research highlighting [...]
Breakthrough Study Reveals Why Damaged Nerves Struggle To Heal
A newly identified molecular mechanism reveals how neurons weigh survival against repair after injury. Scientists at the Icahn School of Medicine at Mount Sinai have identified a molecular switch in neurons that limits the regrowth of [...]
Popular Vitamin B3 Supplements May Help Cancer Cells Survive, Scientists Warn
A new study raises important questions about widely used NAD+ supplements, suggesting that compounds often taken to boost energy and support healthy aging may have unintended consequences in cancer treatment. Millions of Americans take [...]
Scientists Discover Cancer Tumors Are “Addicted” to This Common Antioxidant
Cancer cells may be exploiting a common antioxidant as fuel, revealing a potential weakness that future therapies could target. Cancer cells may be tapping into an unexpected energy source: an antioxidant long associated with [...]
Nanotube injector transfers cytoplasmic contents and organelles between living cells safely
Cells are not isolated units; they continuously exchange proteins, genetic material, and even entire organelles with their neighbors. Intercellular transfer influences how tissues develop, respond to stress, and repair damage. In certain cancers, for [...]
CEO of America’s largest public hospital system is ready to replace radiologists with AI
The chief executive of America’s largest public hospital system says he is prepared to start replacing radiologists with artificial intelligence in some circumstances, once the regulatory landscape catches up. Mitchell H. Katz, MD, president [...]
Our books now available worldwide!
Online Sellers other than Amazon, Routledge, and IOPP Indigo Global Health Care Equivalency in the Age of Nanotechnology, Nanomedicine and Artifcial Intelligence Global Health Care Equivalency In The Age Of Nanotechnology, Nanomedicine And Artificial [...]
Study finds higher heart disease risk in long COVID patients
People with long COVID are at increased risk of developing cardiovascular disease, according to a new study from Karolinska Institutet published in eClinicalMedicine. The results show that the risk of conditions such as cardiac arrhythmias [...]