The COVID-19 pandemic highlighted the need for fast-produced and adaptable vaccines that could be equally distributed around the world. Developing an efficient mRNA vaccine that is effective, thermostable, and has fewer side effects strongly depends on lipid nanoparticles (LNPs) which preserve and deliver mRNA to cells. Lipid nanoparticles appear to be the sole solution to global health issues, and that is why well-known biopharma companies and startups are currently focusing exactly on this.
It might sound easy, but it is not. COVID-19 mRNA vaccines widely used worldwide, such as Comirnaty by Pfizer/BioNTech and Spikevax by Moderna, are proven effective and safe. There are, however, some downsides to current mRNA technologies.
Another issue with today’s COVID-19 mRNA vaccines are the common side effects such as inflammation, fever, and aches. These side effects are caused by lipids which are not fully biodegradable, therefore not disintegrating completely in the body after their work is done. The other challenge of the LNP development is the constant need to adapt to new variants while entrapping bigger payloads to be more efficient.
What can be done to advance LNP technology and the future of mRNA vaccines? As a matter of fact, a lot of scientists are already working on this innovation.
AexeRNA Therapeutics, co-founded by Dr. Drew Weissman, whose research paved the way for the mRNA vaccines, has a proprietary ionizable lipid library. This gives the ability to quickly select the optimal lipids for immunization or therapeutic application. It means that the technology can become more rapidly adjustable to the new variants or applied to other diseases.
The company’s research exemplifies some promising results in mRNA delivery efficiency with controlled distribution to various organs. AexeRNA’s LNP platform is also addressing side effects by lowering toxicity of the system. Their research is dealing with logistics issues by making the vaccine easier to store and transport.
Dr. Pieter Cullis, another world-renowned scientist, has been working on LNP technology for decades. He co-founded and served on the board of more than ten companies, one of which is NanoVation Therapeutics. This company focuses on personalized gene therapeutics. Their pre-clinical studies are focused on the efficacy and biodistribution of LNPs making their research valuable for developing mRNA vaccines.
The other company co-founded by Cullis and led by Dr. Thomas Madden, Acuitas Therapeutics, has an agreement with Pfizer/BioNTech to use its proprietary lipid nanoparticle technology in the development of COVID-19 vaccines. Moreover, the same LNP components can be used in different mRNA vaccines, simplifying their creation for future variants and other diseases.
Last but not least is the revolutionary biopharma startup NeoVac. Using novel lipid nanoparticle technology developed by the company’s co-founder Professor Dan Peer at his Lab at Tel Aviv University, the company aims to overcome existing challenges with the LNPs technology. NeoVac’s in-vivo studies showed fewer side effects due to the fully biodegradable lipids that compose the LNPs. NeoVac’s LNPs have no effect on the liver in fatty liver disease as compared to significant damage by some other vaccines. In addition, their delivery system entraps triple the currently available RNA load and can address multiple variants with multiple payloads simultaneously. This results in higher effectiveness without increasing the dose.
mRNA has proven effective during pandemics and is promising in preventing and treating many other diseases, including cancer. The development of LNPs is crucial for the success of mRNA technologies. The future of global health is in the hands of biopharma companies that are on their way to creating mRNA vaccines with a lower cost of storage and transportation, fewer adverse effects, and faster response to the new variants.
News
The Silent Battle Within: How Your Organs Choose Between Mom and Dad’s Genes
Research reveals that selective expression of maternal or paternal X chromosomes varies by organ, driven by cellular competition. A new study published today (July 26) in Nature Genetics by the Lymphoid Development Group at the MRC [...]
Study identifies genes increasing risk of severe COVID-19
Whether or not a person becomes seriously ill with COVID-19 depends, among other things, on genetic factors. With this in mind, researchers from the University Hospital Bonn (UKB) and the University of Bonn, in [...]
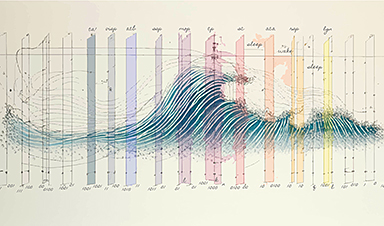
Small regions of the brain can take micro-naps while the rest of the brain is awake and vice versa
Sleep and wake: They're totally distinct states of being that define the boundaries of our daily lives. For years, scientists have measured the difference between these instinctual brain processes by observing brain waves, with [...]
Redefining Consciousness: Small Regions of the Brain Can Take Micro-Naps While the Rest of the Brain Is Awake
The study broadly reveals how fast brain waves, previously overlooked, establish fundamental patterns of sleep and wakefulness. Scientists have developed a new method to analyze sleep and wake states by detecting ultra-fast neuronal activity [...]
AI Reveals Health Secrets Through Facial Temperature Mapping
Researchers have found that different facial temperatures correlate with chronic illnesses like diabetes and high blood pressure, and these can be detected using AI with thermal cameras. They highlight the potential of this technology [...]
Breakthrough in aging research: Blocking IL-11 extends lifespan and improves health in mice
In a recent study published in the journal Nature, a team of researchers used murine models and various pharmacological and genetic approaches to examine whether pro-inflammatory signaling involving interleukin (IL)-11, which activates signaling molecules such [...]
Promise for a universal influenza vaccine: Scientists validate theory using 1918 flu virus
New research led by Oregon Health & Science University reveals a promising approach to developing a universal influenza vaccine—a so-called "one and done" vaccine that confers lifetime immunity against an evolving virus. The study, [...]
New Projects Aim To Pioneer the Future of Neuroscience
One study will investigate the alterations in brain activity at the cellular level caused by psilocybin, the psychoactive substance found in “magic mushrooms.” How do neurons respond to the effects of magic mushrooms? What [...]
Decoding the Decline: Scientific Insights Into Long COVID’s Retreat
Research indicates a significant reduction in long COVID risk, largely due to vaccination and the virus’s evolution. The study analyzes data from over 441,000 veterans, showing lower rates of long COVID among vaccinated individuals compared [...]
Silicon Transformed: A Breakthrough in Laser Nanofabrication
A new method enables precise nanofabrication inside silicon using spatial light modulation and laser pulses, creating advanced nanostructures for potential use in electronics and photonics. Silicon, the cornerstone of modern electronics, photovoltaics, and photonics, [...]
Caught in the actinium: New research could help design better cancer treatments
The element actinium was first discovered at the turn of the 20th century, but even now, nearly 125 years later, researchers still don't have a good grasp on the metal's chemistry. That's because actinium [...]
Innovative Light-Controlled Drugs Could Revolutionize Neuropathic Pain Treatment
A team of researchers from the Institute for Bioengineering of Catalonia (IBEC) has developed light-activated derivatives of the anti-epileptic drug carbamazepine to treat neuropathic pain. Light can be harnessed to target drugs to specific [...]
Green Gold: Turning E-Waste Into a Treasure Trove of Rare Earth Metals
Scientists are developing a process inspired by nature that efficiently recovers europium from old fluorescent lamps. The approach could lead to the long-awaited recycling of rare earth metals. A small molecule that naturally serves [...]
Cambridge Study: AI Chatbots Have an “Empathy Gap,” and It Could Be Dangerous
A new study suggests a framework for “Child Safe AI” in response to recent incidents showing that many children perceive chatbots as quasi-human and reliable. A study has indicated that AI chatbots often exhibit [...]
Nanoparticle-based delivery system could offer treatment for diabetics with rare insulin allergy
Up to 3% of people with diabetes have an allergic reaction to insulin. A team at Forschungszentrum Jülich has now studied a method that could be used to deliver the active substance into the [...]
Nanorobot kills cancer cells in mice with hidden weapon
Researchers at Karolinska Institutet in Sweden have developed nanorobots that kill cancer cells in mice. The robot's weapon is hidden in a nanostructure and is exposed only in the tumor microenvironment, sparing healthy cells. [...]