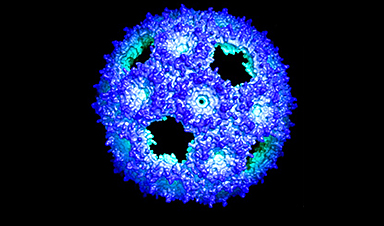
Bacteria across our planet contain nanometer-sized factories that do many different things. Some make nutrients, others isolate toxic materials that could harm the bacteria. We have barely scratched the surface of their functional diversity.
But all share a common exterior, a shell made of protein tiles, that Michigan State University researchers are learning how to manipulate in the lab. This will allow them to build factories of their own design, using the natural building blocks. Indeed, scientists see the structures as a source of new technologies. They are trying to repurpose them to do things they don’t in nature.
In a new study, the lab of Cheryl Kerfeld reports a new genetically engineered shell, based on natural structures and the principles of protein evolution. The new shell is simpler, made of only a single designed protein. It will be easier to work with and, perhaps, even evolve in the lab. The study is published in ACS Synthetic Biology.
Natural shells are made of up to three types of proteins. The most abundant is called BMC-H. Six BMC-H proteins come together to form a hexagon shape to tile the wall.
At some time in evolutionary history, some pairs of BMC-H proteins became joined together, in tandem. Three of these mergers, called BMC-T, join to also form a hexagon shape.
“The two halves of a BMC-T protein can evolve separately while staying next to each other, because they are fused together,” said Bryan Ferlez, a postdoc in the Kerfeld lab. “This evolution allows for diversity in the structures and functions of BMC-T shell proteins, something that we want to recreate by design in the lab.”
To take his fledgling lab to new heights, Liangfang Zhang hatched a plan that he considered brilliant in its simplicity. It involved procedures that many of his peers found a little out there. But if he could make his idea work, it would clear a major hurdle to safely ferry therapies through the body on nanoparticles one-thousandth the width of a human hair.
Yet back in 2010, the young nanoengineer could not convince the National Institutes of Health, the main funder of U.S. biomedical research, to support the project. Zhang applied for funding four or five times over several years, to no avail.
“It felt quite lonely,” he says. “But I just felt this is very unique stuff. And it may become a big thing.”
Pulling funds from other projects and from the start-up package he received to set up his lab at the University of California, San Diego, Zhang did the experiments for his breakthrough paper, published in 2011 in the Proceedings of the National Academy of Sciences. He and coworkers created a new class of nanoparticles, made from carbon-containing polymers, that could slip through blood vessels in a mouse without triggering an immune reaction. While immune responses are important for killing disease-causing pathogens, the same reactions are a nuisance when they clear out molecules made to deliver lifesaving drugs.
Then, instead of just viewing their particles as a drug-delivery system, which most other researchers were focused on, Zhang and his team made a surprising pivot. They repurposed the particles to act as “nanosponges” that trap and remove toxins from the blood. In lab experiments, the nanosponges worked against toxins unleashed by E. coli and some of the harder-to-fight bacteria. Nanosponges also slowed harmful inflammation in mice with a form of rheumatoid arthritis and diverted HIV and Zika from the cells those viruses normally infect, the researchers reported last year.
Image Credit: MSU
News This Week
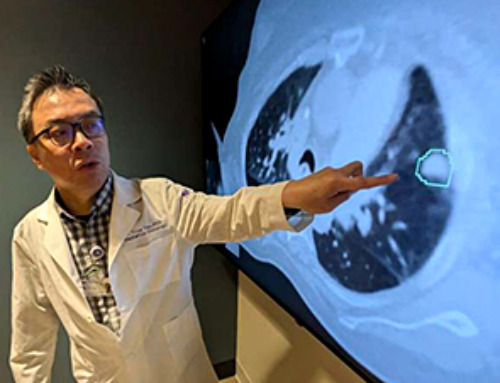
AI matches doctors in mapping lung tumors for radiation therapy
In radiation therapy, precision can save lives. Oncologists must carefully map the size and location of a tumor before delivering high-dose radiation to destroy cancer cells while sparing healthy tissue. But this process, called [...]
Scientists Finally “See” Key Protein That Controls Inflammation
Researchers used advanced microscopy to uncover important protein structures. For the first time, two important protein structures in the human body are being visualized, thanks in part to cutting-edge technology at the University of [...]
AI tool detects 9 types of dementia from a single brain scan
Mayo Clinic researchers have developed a new artificial intelligence (AI) tool that helps clinicians identify brain activity patterns linked to nine types of dementia, including Alzheimer's disease, using a single, widely available scan—a transformative [...]
Is plastic packaging putting more than just food on your plate?
New research reveals that common food packaging and utensils can shed microscopic plastics into our food, prompting urgent calls for stricter testing and updated regulations to protect public health. Beyond microplastics: The analysis intentionally [...]
Aging Spreads Through the Bloodstream
Summary: New research reveals that aging isn’t just a local cellular process—it can spread throughout the body via the bloodstream. A redox-sensitive protein called ReHMGB1, secreted by senescent cells, was found to trigger aging features [...]
AI and nanomedicine find rare biomarkers for prostrate cancer and atherosclerosis
Imagine a stadium packed with 75,000 fans, all wearing green and white jerseys—except one person in a solid green shirt. Finding that person would be tough. That's how hard it is for scientists to [...]
Are Pesticides Breeding the Next Pandemic? Experts Warn of Fungal Superbugs
Fungicides used in agriculture have been linked to an increase in resistance to antifungal drugs in both humans and animals. Fungal infections are on the rise, and two UC Davis infectious disease experts, Dr. George Thompson [...]
Scientists Crack the 500-Million-Year-Old Code That Controls Your Immune System
A collaborative team from Penn Medicine and Penn Engineering has uncovered the mathematical principles behind a 500-million-year-old protein network that determines whether foreign materials are recognized as friend or foe. How does your body [...]
Team discovers how tiny parts of cells stay organized, new insights for blocking cancer growth
A team of international researchers led by scientists at City of Hope provides the most thorough account yet of an elusive target for cancer treatment. Published in Science Advances, the study suggests a complex signaling [...]
Nanomaterials in Ophthalmology: A Review
Eye diseases are becoming more common. In 2020, over 250 million people had mild vision problems, and 295 million experienced moderate to severe ocular conditions. In response, researchers are turning to nanotechnology and nanomaterials—tools that are transforming [...]
Natural Plant Extract Removes up to 90% of Microplastics From Water
Researchers found that natural polymers derived from okra and fenugreek are highly effective at removing microplastics from water. The same sticky substances that make okra slimy and give fenugreek its gel-like texture could help [...]
Instant coffee may damage your eyes, genetic study finds
A new genetic study shows that just one extra cup of instant coffee a day could significantly increase your risk of developing dry AMD, shedding fresh light on how our daily beverage choices may [...]
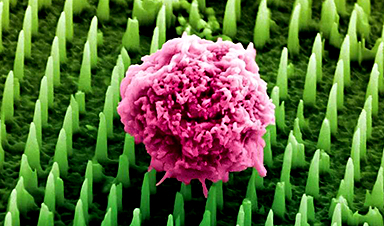
Nanoneedle patch offers painless alternative to traditional cancer biopsies
A patch containing tens of millions of microscopic nanoneedles could soon replace traditional biopsies, scientists have found. The patch offers a painless and less invasive alternative for millions of patients worldwide who undergo biopsies [...]
Small antibodies provide broad protection against SARS coronaviruses
Scientists have discovered a unique class of small antibodies that are strongly protective against a wide range of SARS coronaviruses, including SARS-CoV-1 and numerous early and recent SARS-CoV-2 variants. The unique antibodies target an [...]
Controlling This One Molecule Could Halt Alzheimer’s in Its Tracks
New research identifies the immune molecule STING as a driver of brain damage in Alzheimer’s. A new approach to Alzheimer’s disease has led to an exciting discovery that could help stop the devastating cognitive decline [...]
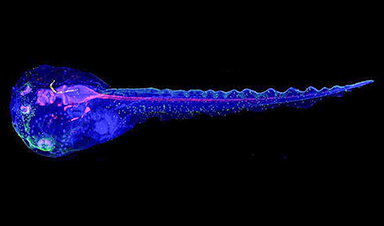
Cyborg tadpoles are helping us learn how brain development starts
How does our brain, which is capable of generating complex thoughts, actions and even self-reflection, grow out of essentially nothing? An experiment in tadpoles, in which an electronic implant was incorporated into a precursor [...]











Leave A Comment