Stress granules play a crucial role in the stress response, arising from the aggregation of non-translating mRNAs and proteins. Although significant knowledge exists about stress granules, the mechanisms behind their mRNA localization remain partially understood. Alterations to mRNA can change the characteristics of the nucleobases, influencing key processes like translation, splicing, and the positioning of specific transcripts.
The researchers show that the RNA modification N4-acetylcytidine (ac4C) on mRNA associates with transcripts enriched in stress granules and that stress granule localized transcripts with ac4C are specifically translationally regulated.
They also show that ac4C on mRNA can mediate the localization of proteins to stress granules. Their results suggest that acetylation of mRNA regulates the localization of both stress-sensitive transcripts and RNA-binding proteins to stress granules and adds to our understanding of the molecular mechanisms responsible for stress granule formation.
Stress Granule Formation and RNA Modifications
Stress granules are membrane-less assemblies of mRNA-protein complexes that arise from mRNAs stuck in translation initiation. RNA-protein complexes are important for their formation and the mechanisms promoting stress granule formation involve both conventional RNA-protein interactions and interactions that encompass intrinsically disordered regions of proteins.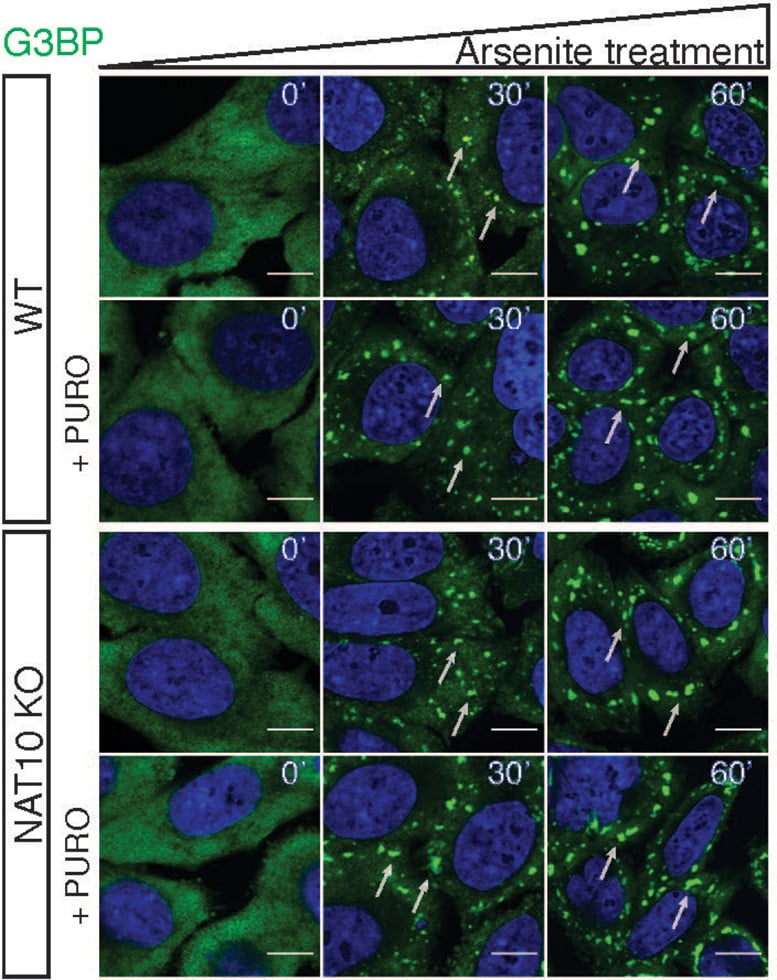
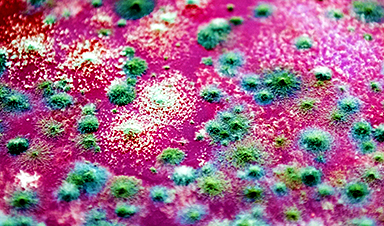
Figure shows stress granule formation after oxidative stress in wild-type cells and cells depleted for the ac4C acetyltransferase enzyme NAT10. Credit: Pavel Kudrin
Stress granules have been extensively studied, and it is well-established that they form when translation initiation is limited a variety of roles for stress granules within the cell have been proposed. While stress granule assembly and disassembly can be regulated by various post-translational modifications the impact of RNA modifications on their formation, dispersal, and function remains largely unclear.
Impact of ac4C on Stress Granules and Cellular Stress Response
The RNA modification N4-acetylcytidine (ac4C) has recently been shown to be deposited on mRNA and regulate translation efficiency. ac4C is conserved through all kingdoms of life and is induced upon several different stresses. ac4C is less abundant than other RNA modifications on mRNA and due to difficulties in precise and quantitative mapping its function and occurrence on mRNA have remained controversial.
The researchers show in their publication that ac4C is enriched in stress granules and that acetylated transcripts are predominantly localized to stress granules in response to oxidative stress, proposing a model where acetylation of RNA can affect mRNA localization to stress granules, in part by affecting the translational release of mRNA from the ribosome, providing new insight into both the function and consequences of mRNA acetylation and mechanism of RNA localization to stress granules.
The findings will promote the understanding of how the cells react to stress and which role RNA modifications play in the process. Both stress and RNA acetylation have implications in disease and their findings could help shed light on relevant molecular pathways that could be targeted in disease.
Reference: “N4-acetylcytidine (ac4C) promotes mRNA localization to stress granules” by Pavel Kudrin, Ankita Singh, David Meierhofer, Anna Kuśnierczyk and Ulf Andersson Vang Ørom, 27 February 2024, EMBO Reports.
DOI: 10.1038/s44319-024-00098-6
The project was led by Ulf A.V. Ørom’s lab at Aarhus University in Denmark, and the study involved collaboration with researchers from the University of Tartu, Norwegian Technical University, and the Max Planck Institute for Molecular Genetics in Berlin.
News
Scientists discover cancer-fighting bacteria that ‘soak up’ forever chemicals in the body
A family of healthy bacteria may help 'soak up' toxic forever chemicals in the body, warding off their cancerous effects. Forever chemicals, also known as PFAS (per- and polyfluoroalkyl substances), are toxic chemicals that [...]
Johns Hopkins Researchers Uncover a New Way To Kill Cancer Cells
A new study reveals that blocking ribosomal RNA production rewires cancer cell behavior and could help treat genetically unstable tumors. Researchers at the Johns Hopkins Kimmel Cancer Center and the Department of Radiation Oncology and Molecular [...]
AI matches doctors in mapping lung tumors for radiation therapy
In radiation therapy, precision can save lives. Oncologists must carefully map the size and location of a tumor before delivering high-dose radiation to destroy cancer cells while sparing healthy tissue. But this process, called [...]
Scientists Finally “See” Key Protein That Controls Inflammation
Researchers used advanced microscopy to uncover important protein structures. For the first time, two important protein structures in the human body are being visualized, thanks in part to cutting-edge technology at the University of [...]
AI tool detects 9 types of dementia from a single brain scan
Mayo Clinic researchers have developed a new artificial intelligence (AI) tool that helps clinicians identify brain activity patterns linked to nine types of dementia, including Alzheimer's disease, using a single, widely available scan—a transformative [...]
Is plastic packaging putting more than just food on your plate?
New research reveals that common food packaging and utensils can shed microscopic plastics into our food, prompting urgent calls for stricter testing and updated regulations to protect public health. Beyond microplastics: The analysis intentionally [...]
Aging Spreads Through the Bloodstream
Summary: New research reveals that aging isn’t just a local cellular process—it can spread throughout the body via the bloodstream. A redox-sensitive protein called ReHMGB1, secreted by senescent cells, was found to trigger aging features [...]
AI and nanomedicine find rare biomarkers for prostrate cancer and atherosclerosis
Imagine a stadium packed with 75,000 fans, all wearing green and white jerseys—except one person in a solid green shirt. Finding that person would be tough. That's how hard it is for scientists to [...]
Are Pesticides Breeding the Next Pandemic? Experts Warn of Fungal Superbugs
Fungicides used in agriculture have been linked to an increase in resistance to antifungal drugs in both humans and animals. Fungal infections are on the rise, and two UC Davis infectious disease experts, Dr. George Thompson [...]
Scientists Crack the 500-Million-Year-Old Code That Controls Your Immune System
A collaborative team from Penn Medicine and Penn Engineering has uncovered the mathematical principles behind a 500-million-year-old protein network that determines whether foreign materials are recognized as friend or foe. How does your body [...]
Team discovers how tiny parts of cells stay organized, new insights for blocking cancer growth
A team of international researchers led by scientists at City of Hope provides the most thorough account yet of an elusive target for cancer treatment. Published in Science Advances, the study suggests a complex signaling [...]
Nanomaterials in Ophthalmology: A Review
Eye diseases are becoming more common. In 2020, over 250 million people had mild vision problems, and 295 million experienced moderate to severe ocular conditions. In response, researchers are turning to nanotechnology and nanomaterials—tools that are transforming [...]
Natural Plant Extract Removes up to 90% of Microplastics From Water
Researchers found that natural polymers derived from okra and fenugreek are highly effective at removing microplastics from water. The same sticky substances that make okra slimy and give fenugreek its gel-like texture could help [...]
Instant coffee may damage your eyes, genetic study finds
A new genetic study shows that just one extra cup of instant coffee a day could significantly increase your risk of developing dry AMD, shedding fresh light on how our daily beverage choices may [...]
Nanoneedle patch offers painless alternative to traditional cancer biopsies
A patch containing tens of millions of microscopic nanoneedles could soon replace traditional biopsies, scientists have found. The patch offers a painless and less invasive alternative for millions of patients worldwide who undergo biopsies [...]
Small antibodies provide broad protection against SARS coronaviruses
Scientists have discovered a unique class of small antibodies that are strongly protective against a wide range of SARS coronaviruses, including SARS-CoV-1 and numerous early and recent SARS-CoV-2 variants. The unique antibodies target an [...]