After a period of falling Covid-19 illness rates, the recent spread of the delta variant of SARS-CoV-2 was a major disappointment and necessitated a reexamination of some previous assumptions. This reconsideration may, at least in part, be a correction to overly optimistic views of what highly effective SARS-CoV-2 vaccines could accomplish. Some observers had hoped the vaccines could eliminate transmission of the virus, the ultimate goal of reaching herd immunity.1 A more likely picture of our future with this virus comes into focus if we examine the well-known infection patterns of another respiratory virus, influenza, both in and outside pandemics. That experience can help us reset expectations and modify goals for dealing with SARS-CoV-2 as it further adapts in global spread.
Early results from the clinical trials and observational studies of mRNA vaccines against SARS-CoV-2 indicated that not only were they highly effective at preventing symptomatic infection, but they were also effective in preventing asymptomatic infection and therefore transmission.2 The basic criterion used for emergency use authorization by the Food and Drug Administration was a standard one: prevention of laboratory-confirmed clinical infection meeting a case definition. The effect on asymptomatic infections was a welcome surprise, because it has been thought that most vaccines for respiratory illnesses, including influenza, are “leaky” — that is, they allow some degree of asymptomatic infection and are better at preventing symptomatic infection.
The initial data on inapparent SARS-CoV-2 infection strengthened the hope that, at a certain level of vaccination, transmission would cease completely. To many of us, this hope appeared overly optimistic, and it seems even more so now; the highly transmissible delta variant causes asymptomatic infections and sometimes illnesses (albeit usually mild) in vaccinated people, probably because of increased growth potential, as well as because of waning immunity, which also involves decreasing IgA antibody levels. Elimination of an illness by means of herd immunity works best when the agent has low transmissibility, and it requires the absence of pockets of susceptible people. Eliminating Covid-19 seemed theoretically possible, because the original 2002 SARS virus ultimately disappeared. That virus, however, did not transmit as well as even the initial strain of SARS-CoV-2. It occurred in limited regions and was characterized by focal spread, including superspreading events. Such a pattern, which was also seen in the early days of SARS-CoV-2, is called “overdispersion” — 10% of cases, for example, may be responsible for 80% of transmission.3 These dynamics explain why there were great differences in antibody prevalence within a given city and spotty global spread early in the pandemic. Overdispersion was thought to be an unstable trait that would disappear, with transmission becoming more uniform and higher overall. That transition appears to have occurred as newer variants take over.
Given the parade of variants, their varying transmissibility, and continuing concern about antigenic changes affecting vaccine protection, I believe it should now be clear that it is not possible to eliminate this virus from the population and that we should develop long-term plans for dealing with it after the unsupportable surges are fully controlled. Pandemic and seasonal influenza provide the most appropriate models to aid in developing strategies going forward.
As with SARS-CoV-2, when a novel pandemic influenza strain appears, its spread can overwhelm the health care system. Waves of infection go through a city in weeks and a country in months, but there is scant evidence that superspreading events occur. Thereafter, the pandemic virus persists as a new seasonal strain, and antigenic changes occur — albeit probably not as quickly as we are seeing with SARS-CoV-2. The new strain joins the other seasonal influenza types and subtypes that reappear each year. The goal of vaccination becomes managing the inevitable outbreaks and reducing the rates of moderate-to-severe illness and death. Preventing mild disease, though important, is less critical.
Readministration of influenza vaccine has become an annual event for much of the population, in response to both waning immunity and the appearance of variants, termed antigenic drift, necessitating updated vaccines. Even when there is no substantial drift, revaccination is recommended because of waning immunity. But antigenic drift is a constant issue and is monitored globally, with vaccine composition updated globally twice a year on the basis of recommendations from a World Health Organization consultation.4 As outlined in the table, various criteria are considered in decisions about which strains to include in vaccines. Vaccine effectiveness against laboratory-confirmed symptomatic infection is never higher than 50 to 60%, and in some years it is much lower. Thus, the value of influenza vaccines, now given to as many as 70% of people in some age groups, lies not in eliminating outbreaks but in reducing them and preventing severe complications.
Though there may be similarities between SARS-CoV-2 and influenza, there are also meaningful differences. The most obvious difference is the efficacy of SARS-CoV-2 vaccines, which is currently much higher than we can achieve with influenza vaccines. Whether that degree of efficacy will continue is one of the many open questions that can only be answered over time. It is clear, however, that revaccination will be necessary, for the same reasons that influenza revaccination is necessary: antigenic variation and waning immunity. Data on the frequency of reinfection with seasonal coronaviruses may not be relevant, but they suggest that protection is relatively short term even after natural infection.5 Revaccination frequency and consequences will need to be determined.
Let us hope that certain problems with the influenza vaccine — such as the failure of vaccination, in some years, to produce the desired increase in protection in previously vaccinated people — do not occur with the SARS-CoV-2 vaccines. Other issues, such as the variant to be targeted by vaccines, will need to be addressed. The successful public–private collaboration in selecting influenza strains offers a model for dealing with such issues. SARS-CoV-2 vaccines will be used globally, and the strain or strains contained in future vaccines will need to be chosen globally, in consultation with the manufacturers.
Most predictions about the shape of the post–Covid-19 world have been inaccurate — a reflection of rapid changes in knowledge. But we can now see a picture emerging in which use of effective vaccines will continue to be critical over the long term. Increases in asymptomatic infections and mild illnesses in vaccinated people will nonetheless continue to be possible, as variants continue to emerge. Counts of hospitalizations and deaths may be more important in monitoring the overall impact than numbers of cases, as long as the vaccines continue to be largely effective at preventing severe illness. The possibility of severe illnesses in a small proportion of vaccinated people does emphasize one of the greatest unmet needs we currently face: continued emphasis on better therapeutics and antiviral agents, which will not be affected by molecular changes in the virus as much as vaccines are….
News
Johns Hopkins Researchers Uncover a New Way To Kill Cancer Cells
A new study reveals that blocking ribosomal RNA production rewires cancer cell behavior and could help treat genetically unstable tumors. Researchers at the Johns Hopkins Kimmel Cancer Center and the Department of Radiation Oncology and Molecular [...]
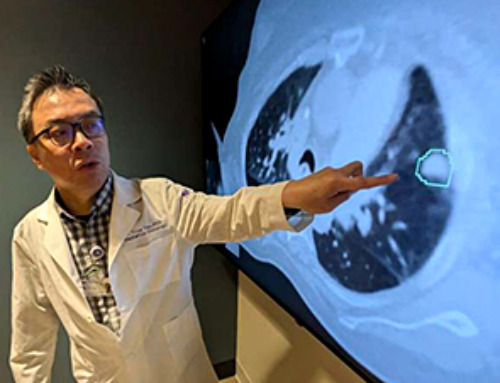
AI matches doctors in mapping lung tumors for radiation therapy
In radiation therapy, precision can save lives. Oncologists must carefully map the size and location of a tumor before delivering high-dose radiation to destroy cancer cells while sparing healthy tissue. But this process, called [...]
Scientists Finally “See” Key Protein That Controls Inflammation
Researchers used advanced microscopy to uncover important protein structures. For the first time, two important protein structures in the human body are being visualized, thanks in part to cutting-edge technology at the University of [...]
AI tool detects 9 types of dementia from a single brain scan
Mayo Clinic researchers have developed a new artificial intelligence (AI) tool that helps clinicians identify brain activity patterns linked to nine types of dementia, including Alzheimer's disease, using a single, widely available scan—a transformative [...]
Is plastic packaging putting more than just food on your plate?
New research reveals that common food packaging and utensils can shed microscopic plastics into our food, prompting urgent calls for stricter testing and updated regulations to protect public health. Beyond microplastics: The analysis intentionally [...]
Aging Spreads Through the Bloodstream
Summary: New research reveals that aging isn’t just a local cellular process—it can spread throughout the body via the bloodstream. A redox-sensitive protein called ReHMGB1, secreted by senescent cells, was found to trigger aging features [...]
AI and nanomedicine find rare biomarkers for prostrate cancer and atherosclerosis
Imagine a stadium packed with 75,000 fans, all wearing green and white jerseys—except one person in a solid green shirt. Finding that person would be tough. That's how hard it is for scientists to [...]
Are Pesticides Breeding the Next Pandemic? Experts Warn of Fungal Superbugs
Fungicides used in agriculture have been linked to an increase in resistance to antifungal drugs in both humans and animals. Fungal infections are on the rise, and two UC Davis infectious disease experts, Dr. George Thompson [...]
Scientists Crack the 500-Million-Year-Old Code That Controls Your Immune System
A collaborative team from Penn Medicine and Penn Engineering has uncovered the mathematical principles behind a 500-million-year-old protein network that determines whether foreign materials are recognized as friend or foe. How does your body [...]
Team discovers how tiny parts of cells stay organized, new insights for blocking cancer growth
A team of international researchers led by scientists at City of Hope provides the most thorough account yet of an elusive target for cancer treatment. Published in Science Advances, the study suggests a complex signaling [...]
Nanomaterials in Ophthalmology: A Review
Eye diseases are becoming more common. In 2020, over 250 million people had mild vision problems, and 295 million experienced moderate to severe ocular conditions. In response, researchers are turning to nanotechnology and nanomaterials—tools that are transforming [...]
Natural Plant Extract Removes up to 90% of Microplastics From Water
Researchers found that natural polymers derived from okra and fenugreek are highly effective at removing microplastics from water. The same sticky substances that make okra slimy and give fenugreek its gel-like texture could help [...]
Instant coffee may damage your eyes, genetic study finds
A new genetic study shows that just one extra cup of instant coffee a day could significantly increase your risk of developing dry AMD, shedding fresh light on how our daily beverage choices may [...]
Nanoneedle patch offers painless alternative to traditional cancer biopsies
A patch containing tens of millions of microscopic nanoneedles could soon replace traditional biopsies, scientists have found. The patch offers a painless and less invasive alternative for millions of patients worldwide who undergo biopsies [...]
Small antibodies provide broad protection against SARS coronaviruses
Scientists have discovered a unique class of small antibodies that are strongly protective against a wide range of SARS coronaviruses, including SARS-CoV-1 and numerous early and recent SARS-CoV-2 variants. The unique antibodies target an [...]
Controlling This One Molecule Could Halt Alzheimer’s in Its Tracks
New research identifies the immune molecule STING as a driver of brain damage in Alzheimer’s. A new approach to Alzheimer’s disease has led to an exciting discovery that could help stop the devastating cognitive decline [...]