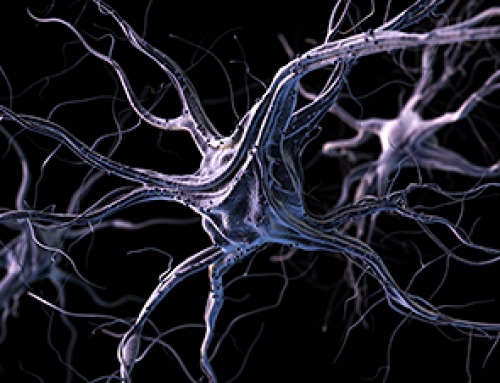
Neurological symptoms are apparently not a result of SARS-CoV-2 infection of the brain.
Scientists still are not sure how neurological symptoms arise in COVID-19. Is it because SARS-CoV-2 infects the brain? Or are these symptoms the result of inflammation in the rest of the body? A study by Charité – Universitätsmedizin Berlin has now produced evidence to support the latter theory. It was published today in the journal Nature Neuroscience.
Headaches, memory problems, and fatigue are just some of the neurological impacts that arise during coronavirus infection and can last well beyond the acute period. Even early on in the pandemic, researchers surmised that direct infection of the brain could be the cause.
"We took that as our hypothesis at the start, too. But so far, there has been no clear evidence that the coronavirus can persist in the brain, let alone proliferate," explains Dr. Helena Radbruch, head of the Chronic Neuroinflammation working group at the Department of Neuropathology at Charité. "For that, we would have needed to find evidence of intact virus particles in the brain, for example. Instead, the indications that the coronavirus could infect the brain come from indirect testing methods, so they aren't entirely conclusive."
According to a second hypothesis, the neurological symptoms would instead be a kind of side effect of the strong immune response the body deploys to defend against the virus. Past studies have produced indications that this might be the case. The current Charité study now bolsters this theory with detailed molecular biology and anatomical results from autopsies.
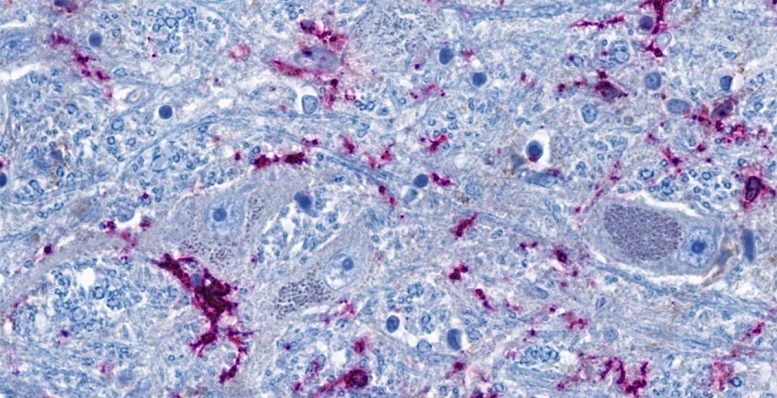
Cross-section of the brainstem: Neurons (blue-gray) are in close contact with immune cells (purple). The threadlike blue structures are extensions of the neurons, which can reach all the way into distant organs in the form of nerve fibers. According to the study, the immune cells and neurons in the brainstem can be activated directly via the nerve fibers as a result of inflammation in the lungs. Credit: © Charité | Jenny Meinhardt
No Signs of Direct Infection of the Brain
For the study, the team of researchers analyzed various areas of the brain in 21 people who died in hospital settings, typically in an ICU, due to severe coronavirus infection. For comparison, the researchers studied nine patients who died of other causes after treatment in intensive care. First, they looked to see whether the tissue showed any visible changes and hunted for any indication of coronavirus. Then they conducted a detailed analysis of genes and proteins to identify the specific processes that had taken place inside individual cells.
Like other teams of researchers before them, the Charité scientists found coronavirus genetic material in the brain in some cases. "But we didn't find neurons infected with SARS-CoV-2," Radbruch notes. "We assume that immune cells absorbed the virus in the body and then traveled to the brain. They're still carrying the virus, but it doesn't infect cells of the brain. So coronavirus has invaded other cells in the body, but not the brain itself."
Brain Reacts to Inflammation in the Body
Still, the researchers did note striking changes in molecular processes in some cells of the brain in those infected with COVID-19: For example, the cells ramped up the interferon signaling pathway, which is typically activated in the course of a viral infection.
"Some neurons evidently react to the inflammation in the rest of the body," says Prof. Christian Conrad, head of the Intelligent Imaging working group at the Berlin Institute of Health at Charité (BIH) and one of the principal investigators in the study, along with Radbruch. "This molecular reaction could be a good explanation for the neurological symptoms we see in COVID-19 patients. For example, neurotransmitters emitted by these cells in the brainstem could cause fatigue. That's because the brainstem is home to groups of cells that control drive, motivation, and mood."
The reactive nerve cells were found primarily in what are known as the nuclei of the vagus nerve. These are nerve cells located in the brainstem that extend all the way to organs such as the lungs, intestine, and heart. "In simplified terms, our interpretation of our data is that the vagus nerve 'senses' the inflammatory response in different organs of the body and reacts to it in the brainstem – without there being any actual infection of brain tissue," Radbruch explains. "Through this mechanism, the inflammation does spread from the body to the brain in a way, which can disrupt brain function."
Limited-Time Reaction
The neurons' reaction to the inflammation is temporary, as shown by a comparison of people who died during an acute coronavirus infection with those who died at least two weeks afterward. The molecular changes are most evident during the acute infection phase, but they do normalize again afterward – at least in the vast majority of cases.
"We think it's possible that if the inflammation becomes chronic, that could be what causes the neurological symptoms often observed in long COVID in some people," Conrad says. To follow up on this suspicion, the team of researchers is now planning to study the molecular signatures in the cerebral fluid of long COVID patients in greater detail.
Reference: "Proteomic and transcriptomic profiling of brainstem, cerebellum and olfactory tissues in early- and late-phase COVID-19" by Josefine Radke, Jenny Meinhardt, Tom Aschman, Robert Lorenz Chua, Vadim Farztdinov, Sören Lukassen, Foo Wei Ten, Ekaterina Friebel, Naveed Ishaque, Jonas Franz, Valerie Helena Huhle, Ronja Mothes, Kristin Peters, Carolina Thomas, Shirin Schneeberger, Elisa Schumann, Leona Kawelke, Julia Jünger, Viktor Horst, Simon Streit, Regina von Manitius, Péter Körtvélyessy, Stefan Vielhaber, Dirk Reinhold, Anja E. Hauser, Anja Osterloh, Philipp Enghard, Jana Ihlow, Sefer Elezkurtaj, David Horst, Florian Kurth, Marcel A. Müller, Nils C. Gassen, Julia Melchert, Katharina Jechow, Bernd Timmermann, Camila Fernandez-Zapata, Chotima Böttcher, Werner Stenzel, Elke Krüger, Markus Landthaler, Emanuel Wyler, Victor Corman, Christine Stadelmann, Markus Ralser, Roland Eils, Frank L. Heppner, Michael Mülleder, Christian Conrad and Helena Radbruch, 16 February 2024, Nature Neuroscience.
DOI: 10.1038/s41593-024-01573-y
News
Cancer Mystery Solved: Scientists Discover How Melanoma Becomes “Immortal”
Scientists have uncovered a previously overlooked mechanism that may help melanoma cells become effectively “immortal.” Cancer cells face a major problem before they can become deadly: They have to figure out how to stop [...]
How Visual Neurons Organize Thousands of Synaptic Inputs
Summary: A new study uncovered the organizational rules that determine how neurons in the primary visual cortex process information. By imaging both the cell bodies (soma) and the individual synapses (on dendritic spines) of [...]
Scientists Just Found a Surprising Way To Destroy “Forever Chemicals”
Scientists have uncovered a new mechanism that may help break down highly persistent PFAS pollutants. PFAS have earned the nickname “forever chemicals” for a reason. These industrial compounds are so chemically durable that they [...]
Scientists Discover Cheap Material That Kills Deadly Superbugs
A new sulfur-rich antimicrobial polymer shows strong effectiveness against fungal and bacterial pathogens and may offer an affordable solution to antimicrobial resistance. Antimicrobial resistance is creating growing challenges for both healthcare and food production, [...]
What to Know About Cicada, or BA.3.2, the Latest SARS-CoV-2 Variant Under Monitoring
Like periodical cicadas, the insects for which it is nicknamed, SARS-CoV-2 Omicron subvariant BA.3.2 is only just beginning to emerge after lying low for an extended period since it first appeared. Although it was [...]
Scientists Say This Simple Supplement May Actually Reverse Heart Disease
Scientists in Japan say a common supplement may actually help “unclog” certain diseased heart arteries from the inside out. A simple food supplement sold in Japan may have helped reverse a dangerous form of [...]
New breakthrough against radiation: Korean Scientists create revolutionary shield with nanotechnology
Korean Scientists develop new nanotechnology material capable of reducing radiation impacts in space missions, hospitals, and power plants. The search for more efficient protection technologies in extreme environments has just gained an important advance. Korean [...]
Scientists Just Discovered the Hidden Trick That Keeps Your Cells Alive
A strange bead-like motion inside cells may be the secret to keeping their DNA—and health—in balance. Mitochondria are often described as the power plants of the cell because they produce the energy cells need [...]
Scientists Discover Stem Cells That Could Regrow Teeth and Bone
Scientists just uncovered the cellular “blueprint” that could one day let us regrow real teeth. Researchers at Science Tokyo have uncovered two distinct stem cell lineages that play a central role in forming tooth [...]
Scientists Uncover Fatal Weakness in “Zombie Cells” Linked to Cancer
A newly identified weakness in “zombie” cells may open the door to more precise cancer treatments by turning their own survival strategy against them. A new class of drugs takes advantage of a recently [...]
Bowel and Ovarian Cancers Are Dramatically Rising in Young Adults, Scientists Aren’t Sure Why
Cancer incidence is increasing, especially among younger adults, and current risk factors don’t fully account for the trend. Scientists suggest other underlying causes may be contributing. Cancer patterns in England are shifting in a [...]
New Immune Pathway Could Supercharge mRNA Cancer Vaccines
A surprising backup system in the immune response to mRNA vaccines may hold the key to more effective cancer treatments. The arrival of mRNA vaccines against SARS-CoV-2 in 2020 marked a turning point in the COVID-19 pandemic. Today, [...]
Scientists Discover “Molecular Switch” That Fuels Alzheimer’s Brain Inflammation
A newly identified trigger of brain inflammation could offer a fresh target for slowing Alzheimer’s progression. The brain has its own built-in immune system that identifies threats and responds to them. In Alzheimer’s disease, growing evidence [...]
Molecular Manufacturing: The Future of Nanomedicine – New book from NanoappsMedical Inc.
This book explores the revolutionary potential of atomically precise manufacturing technologies to transform global healthcare, as well as practically every other sector across society. This forward-thinking volume examines how envisaged Factory@Home systems might enable the cost-effective [...]
Forgotten Medicinal Plant Shows Promise in Fighting Dangerous Superbugs
A traditional medicinal plant, tormentil, shows promise against antibiotic-resistant bacteria in laboratory tests. Its compounds work by limiting bacterial growth and boosting antibiotic performance. Before the development of modern antibiotics, plant-based remedies were commonly [...]
NanoMedical Brain/Cloud Interface – Explorations and Implications. A new book from Frank Boehm
New book from Frank Boehm, NanoappsMedical Inc Founder: This book explores the future hypothetical possibility that the cerebral cortex of the human brain might be seamlessly, safely, and securely connected with the Cloud via [...]