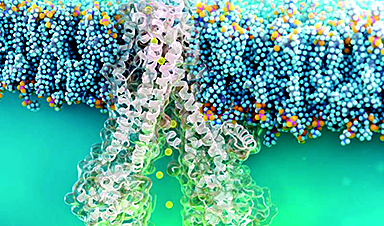
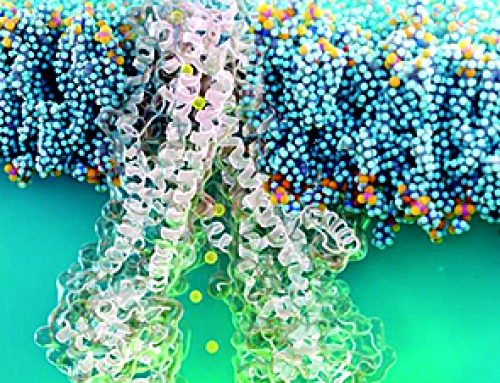
| Diseased cells such as metastatic cancer cells have markedly different mechanical properties that can be used to improve targeted drug uptake, according to a team of researchers at Penn State. | |
| Many labs around the world are developing nanoparticle-based drug delivery systems to selectively target tumors. They rely on a key-and-lock system in which protein keys on the surface of the nanoparticle click into the locks of a highly expressed protein on the surface of the cancer cell. The cell membrane then wraps around the nanoparticle and ingests it. If enough of the nanoparticles and their drug cargo is ingested, the cancer cell will die. | |
| The adhesive force of the lock and key is what drives the nanoparticle into the cell, says Sulin Zhang, professor of engineering science and mechanics. “It is almost universal that whenever there is a driving force for a process, there always is a resistive force. Here, the driving force is biochemical – the protein-protein interaction.” | |
| The resistive force is mechanical, the energy cost of the membrane wrapping around the nanoparticle. Until now, bioengineers only considered the driving force and designed nanoparticles to optimize the chemical interactions, a targeting strategy called “chemotargeting.” Zhang believes they should also take into account the mechanics of the cells to design nanoparticles to achieve enhanced targeting, which forms a new targeting strategy called “mechanotargeting.” |
Image Credit: Zhang lab/vecteezy.com
News This Week
Nanobody repairs misfolded CFTR inside cells, boosting function in cystic fibrosis
A tiny antibody component could fundamentally transform the treatment of cystic fibrosis: For the first time, researchers have succeeded in developing a so-called nanobody that penetrates directly into human cells and can repair the [...]
20-Year Study Finds Daily Multivitamins Don’t Extend Lifespan
A large, decades-long study of over 390,000 U.S. adults challenges a widespread assumption about daily multivitamins. Multivitamins are a daily habit for millions of Americans, often taken with the expectation that they will extend [...]
Novel Investment Paradigms for Regenerative Healthcare Ecosystems
Introduction The transition toward regenerative healthcare ecosystems—anchored in wellness optimization, disease prevention, eradication strategies, and healthy longevity—necessitates a structural reconfiguration of capital architectures, governance models, and incentive design. Regenerative healthcare, by definition, transcends episodic [...]
What If Consciousness Exists Beyond Your Brain
Scientists still don’t know how consciousness emerges from the brain. New ideas suggest it may not emerge at all, but instead be a basic feature of reality. Is consciousness produced by the brain, or [...]
Scientists Discover Way To Treat Lung Cancer and Its Deadly Side Effect Together
A new approach using lipid nanoparticles to deliver genetic material is showing promise in tackling two major challenges in lung cancer at once.Researchers at Oregon State University have designed a new way to tackle two of [...]
Saunas Activate Your Immune System
A brief sauna session may quietly mobilize the immune system. A sauna session may do more than raise your heart rate and body temperature. A new study from Finland found that it also briefly [...]
Why music from your youth still has such an intense effect years later: A psychological perspective
You're driving, and suddenly a familiar song fills the air. Before you even know it, a wave of emotions comes over you – not just memories, but a deep, almost physical feeling. This powerful [...]
AI to antibody in days: breaking the wet lab bottleneck via high-throughput integration
The role of artificial intelligence (AI) in drug design has fundamentally shifted from a speculative tool to a central pillar of pharmaceutical research and development (R&D). Sino Biological plays a critical role in this [...]







Leave A Comment